
Neuroprotective Activity of Clematis mandshurica
Abstract
Oxidative stress is widely recognized as a central driver in the progression of neurodegenerative diseases, where it contributes to neuronal apoptosis through diverse intracellular signaling mechanisms. In this study, we examined the neuroprotective potential of Clematis mandshurica, a traditional Korean medicinal herb historically used to alleviate conditions such as urethritis, carbuncles, and certain malignancies. To model oxidative injury, neuronal cells were exposed to excessive glutamate, a treatment known to elevate reactive oxygen species (ROS), trigger intracellular calcium (Ca2+) overload, impair mitochondrial membrane potential, and diminish the activities of key glutathione-dependent enzymes, including glutathione reductase (GR) and glutathione peroxidase (GPx). Treatment with C. mandshurica extract effectively counteracted these glutamate-induced alterations, as demonstrated by improved cell viability in MTT assays, suppression of ROS and Ca2+ accumulation, preservation of mitochondrial function, and restoration of GR and GPx activity. Collectively, these findings suggest that C. mandshurica exerts robust cytoprotective actions and holds promise as a potential therapeutic agent for neurodegenerative disorders characterized by oxidative stress, including Alzheimer’s disease.
Keywords:
Oxidative stress, Neuroprotection, Clematis mandshurica, Glutamate toxicity, Mitochondrial dysfunction, Alzheimer’s diseaseAlzheimer’s disease (AD) is the most common form of dementia and constitutes a progressive neurodegenerative syndrome marked by a continual decline in cognitive capacity and memory function.1) As life expectancy increases and medical care improves, the incidence of AD in individuals aged 65 years and older has grown substantially, becoming a major public health and societal concern. Several well-established risk factors contribute to AD development, including advancing age, genetic susceptibility, and family history.2) Clinically, patients typically exhibit a gradual progression of cognitive impairment, most notably involving deficits in memory and higher-order cognitive processes.
From a pathological standpoint, Alzheimer’s disease (AD) is characterized by the accumulation of amyloid-β (Aβ) plaques, the development of neurofibrillary tangles (NFTs), and widespread neuronal degeneration driven in part by oxidative stress and cholinergic dysfunction.3) Aβ plaques form through the progressive aggregation of β-amyloid peptides, while NFTs arise from the abnormal hyperphosphorylation and aggregation of tau, a microtubule-associated protein. In addition, inflammatory responses elicited around these pathological lesions further compromise neuronal stability and contribute to ongoing neurodegeneration.4)
Oxidative stress is widely acknowledged as a central pathogenic mechanism in Alzheimer’s disease (AD).5) Cellular events such as lipid peroxidation, free radical accumulation, protein oxidation, and DNA damage collectively contribute to neuronal dysfunction and necrosis within the central nervous system (CNS), thereby aggravating disease progression. Among the major mediators of oxidative injury are reactive oxygen species (ROS), including hydroxyl radicals (OH•), superoxide anions (O2⁻), and hydrogen peroxide (H2O2). Excessive intracellular calcium (Ca2+) influx—often resulting from overstimulation of NMDA receptors—further intensifies ROS generation and heightens neuronal susceptibility to damage.6)
Currently available pharmacological therapies for Alzheimer’s disease (AD) largely target symptomatic relief and primarily include acetylcholinesterase (AChE) inhibitors such as donepezil, galantamine, and tacrine as well as NMDA receptor antagonists. Although these agents offer modest improvements in cognitive function, their clinical utility is often limited by adverse effects, including appetite suppression, nausea, vomiting, and diarrhea.7) As a result, growing interest has been directed toward medicinal plants with neuroprotective properties, such as Ginkgo biloba, Salvia officinalis, Melissa officinalis, and Papaver somniferum, which have been reported to exert cognitive-enhancing effects while exhibiting fewer side effects in patients with AD.8)
Clematis mandshurica (family Ranunculaceae), commonly referred to as “Weilingxian,” is a traditional medicinal plant that has long been utilized for its anti-inflammatory, antitumor, analgesic, and diuretic activities.9-11) The rhizomes and roots of the plant are distinguished by their brown, fibrous surfaces with prominent longitudinal wrinkles and are known to contain a diverse array of bioactive constituents, including anemonin, anemonol, sterols, saponins, lactones, and various amino acids.12,13) A growing body of evidence indicates that C. mandshurica exhibits protective effects in experimental models of osteoarthritis, polyarthritis, and streptozotocin-induced diabetes, underscoring its broad pharmacological potential and therapeutic relevance.
In the CNS, glutamate serves as the primary excitatory neurotransmitter; however, excessive glutamate induces oxidative stress and apoptosis by inhibiting cystine uptake and by modulating MAPK signaling pathways, including JNK, p38, and ERK.14,15) The HT22 cell line, derived from mouse hippocampal neurons, is widely employed as an in vitro model to investigate glutamate-induced neurotoxicity and oxidative stress-associated mechanisms.16)
In this study, we examined the neuroprotective effects of C. mandshurica extract against glutamate-induced toxicity in mouse hippocampal HT22 cells. Excess glutamate was used to induce oxidative stress and apoptotic signaling, allowing evaluation of the extract’s protective capacity. Cellular and biochemical assessments were conducted to determine whether the extract could improve neuronal survival and modulate key oxidative stress markers. These analyses enabled us to characterize the potential of C. mandshurica as a neuro-protective agent under glutamate-driven stress conditions.
Experimental
Plant Materials and Extract Preparation − The roots of Clematis mandshurica were sourced from the Kyungdong Traditional Herbal Market in Seoul, Korea. A voucher specimen (CJ141M) was authenticated and deposited in the Natural Products Laboratory of Kangwon National University for reference and quality control. The air-dried roots (6.0 kg) were extracted with 80% methanol using an ultrasonication-assisted procedure. The extraction was carried out three times for 90 minutes each, at a solvent-to-material ratio of 1 L per kilogram. The pooled methanolic extracts were then concentrated under reduced pressure and subsequently dried, yielding a crude extract used for subsequent experimental analyses.
Cell Viability − Mouse hippocampal HT22 cells were obtained from Seoul National University (Seoul, Korea) and maintained under standardized culture conditions. Cells were grown in Dulbecco’s Modified Eagle’s Medium (DMEM; Gibco, USA) supplemented with 10% fetal bovine serum (FBS), 1% penicillin–streptomycin, 2 mg/mL NaHCO₃, and 15 mM HEPES. Cultures were incubated at 37°C in a humidified atmosphere containing 5% CO2.
Cell viability was evaluated using the MTT assay. HT22 cells were seeded at 2.0 × 104 cells per well in 48-well plates and allowed to adhere for 24 h. Samples and trolox (positive control) were administered 1 h prior to the addition of 2 mM glutamate to induce cytotoxicity. Following treatment, MTT solution (1 mg/mL) was added, and cells were incubated for an additional 3 h. Formazan crystals formed in viable cells were solubilized with dimethyl sulfoxide (DMSO), and absorbance was recorded at 570 nm using a microplate reader. Cell viability was expressed relative to untreated control cells. Glutamate, MTT, DMSO, and trolox were sourced from Sigma-Aldrich (USA). Neuroprotective activity was calculated as relative protection (%) according to the following equation:
Relative protection (%) = [(OD of glutamate-treated with sample group − OD of glutamate-only group) / (OD of control group − OD of glutamate-only group)] × 100.
Measurement of Intracellular ROS Levels − Intracellular reactive oxygen species (ROS) levels were assessed using the fluorescent probe 2’,7’-dichlorofluorescein diacetate (DCF-DA). HT22 cells were seeded into 48-well plates and co-treated with the test sample and 2 mM glutamate for 8 h. After incubation, cells were gently washed with PBS and incubated with 10 µM DCF-DA prepared in Hanks’ balanced salt solution for 30 min in the dark to prevent photobeaching. Following staining, the cells were washed again with PBS and lysed with 1% Triton X-100 in PBS for 10 min at 37°C. Fluorescence intensity, indicative of intracellular ROS accumulation, was measured using a microplate reader at excitation and emission wavelengths of 490 nm and 525 nm, respectively.
Measurement of Intracellular Ca2+ Levels − Cytosolic calcium (Ca2+) levels in HT22 cells were quantified using the calcium-sensitive fluorescent indicator Fura-2 AM. Cells were seeded into 48-well plates and allowed to attach for 24 h prior to experimentation. The samples, glutamate, and 2 µM Fura-2 AM were then added simultaneously, followed by a 2 h incubation period. After treatment, cells were rinsed gently with PBS and lysed using 1% Triton X-100 in PBS for 10 min at 37°C. Fluorescence intensity was subsequently measured with a microplate reader at excitation and emission wavelengths of 340 nm/380 nm and 535 nm, respectively, providing an index of intracellular Ca2+ accumulation.
Measurement of Mitochondrial Membrane Potential − Mitochondrial membrane potential (ΔΨm) was evaluated using the cationic fluorescent dye rhodamine 123 (Rho123), which selectively accumulates within polarized mitochondria. HT22 cells were seeded into 48-well plates and allowed to adhere for 24 h before treatment. Cells were then exposed to the test sample together with 2 mM glutamate for 24 h to induce mitochondrial dysfunction. Following treatment, the medium was removed and cells were incubated with Rho123 for 15 min at 37°C in the dark to permit mitochondrial uptake of the dye. After staining, cells were washed with PBS to eliminate excess dye, and fluorescence was measured immediately using a microplate reader at excitation and emission wavelengths of 488 nm and 520 nm, respectively. A decrease in fluorescence intensity was interpreted as a loss of ΔΨm, whereas preservation or recovery of fluorescence indicated a protective effect of the sample on mitochondrial membrane integrity.
Measurement of Glutathione, Glutathione Reductase and Glutathione Peroxides − HT22 cells were seeded into 6-well plates and treated with the test sample together with 2 mM glutamate for 24 h. After incubation, the cells were collected and centrifuged at 3000 × g for 30 min at 4°C, and the resulting supernatant was used to evaluate antioxidant enzyme activities, including total glutathione (GSH), glutathione reductase (GR), and glutathione peroxidase (GPx).
Total GSH content was measured based on its reaction with DTNB (5,5′-dithiobis-2-nitrobenzoic acid). The supernatant was combined with 0.3 mM NADPH and 0.5 mM DTNB and incubated for 5 min at 35°C, followed by the addition of glutathione reductase (5 U/mL). The formation of the yellow TNB chromophore was monitored spectro-photometrically at 412 nm.
GR activity was determined by quantifying the NADPH-dependent reduction of oxidized glutathione (GSSG). The assay mixture consisted of the sample supernatant, 1 mM GSSG, and phosphate buffer containing 0.1 mM NADPH. The decrease in absorbance at 340 nm was recorded at 15-s intervals over a 120-s period.
GPx activity was analyzed using a coupled assay in the presence of glutathione reductase. The reaction mixture contained 1 mM GSH, 0.4 mM NADPH, 0.2 mM H2O2, and 1 U/mL glutathione reductase. GPx activity was calculated by measuring the rate of NADPH oxidation, monitored by the decrease in absorbance at 340 nm.
Statistical Analysis − All data are presented as mean ± standard deviation (SD). Statistical differences among experimental groups were evaluated using one-way analysis of variance (ANOVA), followed by Tukey’s post hoc test for multiple comparisons. Data processing and statistical analyses were performed using IBM SPSS Statistics (version 26) and Microsoft Excel. Significance thresholds were set at p < 0.05, p < 0.01, and p < 0.001, which indicate increasing levels of statistical significance between treatment groups.
Results and Discussion
To investigate whether C. mandshurica exhibits protective effects against glutamate-induced neuronal injury, The C. mandshurica extract itself did not exhibit any neurotoxicity in HT22 cells (data not shown). HT22 cells were pretreated with the extract and then exposed to glutamate. As expected, glutamate markedly decreased cell viability, confirming successful induction of excitotoxic stress. Pretreatment with C. mandshurica extract, however, significantly improved cell survival in a concentration-dependent manner. At 100 μg/mL, the extract restored cell viability to a relative protective value of 54.11 ± 3.6%, indicating strong neuroprotection (Fig. 1).
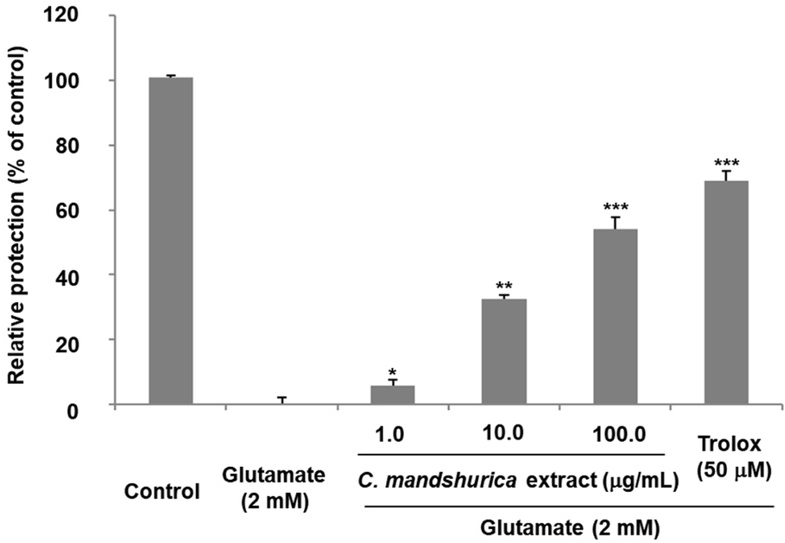
Neuroprotective effects of C. mandshurica extract against glutamate-induced cytotoxicity in HT22 cells. HT22 cells were pretreated with C. mandshurica extract (10, 50, and 100 μg/mL) and subsequently exposed to glutamate (2 mM) for 24 h. Cell viability was assessed, and data are presented as the mean ± SD from three independent experiments. *p < 0.05, **p < 0.01, ***p < 0.001 compared with glutamate-treated control cells.
These results suggest that C. mandshurica counteracts glutamate-induced cytotoxicity, potentially through antioxidant activity, regulation of intracellular signaling, and stabilization of metabolic pathways associated with cell survival. Similar protective patterns have been reported for other glutamate-modulating or antioxidant plant extracts, further supporting the therapeutic potential of C. mandshurica in glutamate-related neurodegeneration.17)
Glutamate toxicity is closely related to excessive production of reactive oxygen species (ROS), which aggravates oxidative stress and triggers apoptotic pathways.18) Consistent with this mechanism, glutamate treatment markedly elevated intracellular ROS levels in HT22 cells. Using H₂DCF-DA fluorescence, we confirmed a substantial increase in ROS following glutamate exposure. Importantly, C. mandshurica pretreatment significantly suppressed ROS accumulation. At concentrations of 10 μg/ml and 100 μg/ml, ROS levels were reduced to 122.98 ± 2.43% and 110.23 ± 3.56% of control, respectively (Fig. 2). This attenuation of oxidative stress indicates that C. mandshurica possesses active compounds capable of acting either as direct free-radical scavengers or as modulators of endogenous antioxidant pathways. Considering that elevated ROS is one of the earliest events in glutamate-induced apoptosis, the ROS-suppressing activity of C. mandshurica is likely a major contributor to its overall neuroprotective effect. These findings align with previous studies reporting that natural products with phenolic or flavonoid components often exert potent ROS-inhibitory actions in neuronal models.
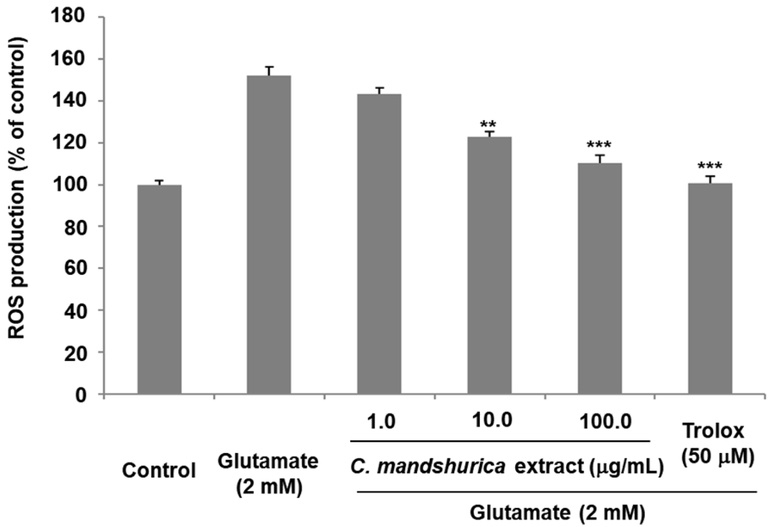
The effects of C. mandshurica extract on glutamate-induced ROS generation in HT22 cells. Intracellular ROS levels were measured following pretreatment with C. mandshurica extract and subsequent exposure to glutamate. ROS production is presented as a percentage relative to control cells. Data represent the mean ± SD of three independent experiments. *p < 0.05, **p < 0.01, ***p < 0.001 compared with glutamate-treated cells.
Intracellular Ca2+ overload is a well-established consequence of glutamate excitotoxicity that amplifies oxidative stress, disrupts mitochondrial function, and triggers apoptosis.19) In this study, glutamate treatment significantly elevated intracellular Ca2+ levels in HT22 cells, confirming excitotoxic stimulation. Pretreatment with C. mandshurica markedly reduced Ca2+ accumulation, lowering Ca2+ levels to 105.78 ± 3.21% at 100 μg/ml (Fig. 3). This suppression of Ca2+ influx suggests that C. mandshurica may interfere with glutamate-induced Ca2+ dysregulation by modulating NMDA receptor-mediated Ca2+ entry, stabilizing calcium-buffering capacity, or reducing oxidative damage that exacerbates Ca2+ flux. Restoration of Ca2+ homeostasis is critical for neuronal survival, indicating that C. mandshurica plays a key role in protecting neurons from Ca2+-induced apoptosis.20)
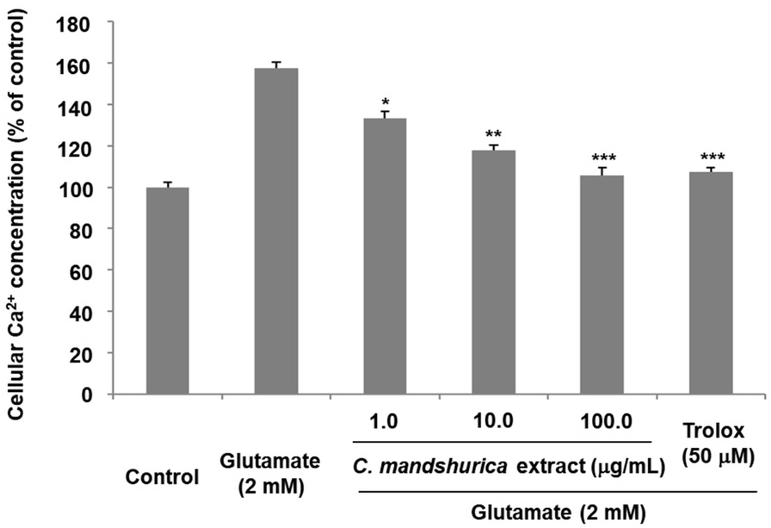
The Protective effects of C. mandshurica extract against glutamate-induced Ca2+ influx in HT22 cells. HT22 cells were pretreated with C. mandshurica extract prior to glutamate exposure, and intracellular Ca2+ levels were quantified. Data are expressed as a percentage relative to control cells. Values represent the mean ± SD of three independent experiments. *p < 0.05, **p < 0.01, ***p < 0.001 compared with glutamate-treated cells.
Mitochondria are central regulators of neuronal survival, and collapse of mitochondrial membrane potential (ΔΨm) is a hallmark of early apoptosis.21) Consistent with previous studies, glutamate treatment resulted in a significant decrease in ΔΨm, reflecting mitochondrial dysfunction and depolarization.22) C. mandshurica pretreatment, however, effectively restored mitochondrial membrane integrity. At 100 μg/ml, ΔΨm increased to 86.72 ± 2.6% of the control, indicating a strong protective effect (Fig. 4). Maintenance of ΔΨm suggests that C. mandshurica either prevents mitochondrial damage or enhances mitochondrial resilience during glutamate-induced stress. Stabilizing mitochondrial function is critical because mitochondrial depolarization leads to ATP depletion, ROS overproduction, and release of pro-apoptotic factors such as cytochrome c. Thus, recovery of ΔΨm highlights a key mechanism by which C. mandshurica prevents excitotoxic neuronal death.
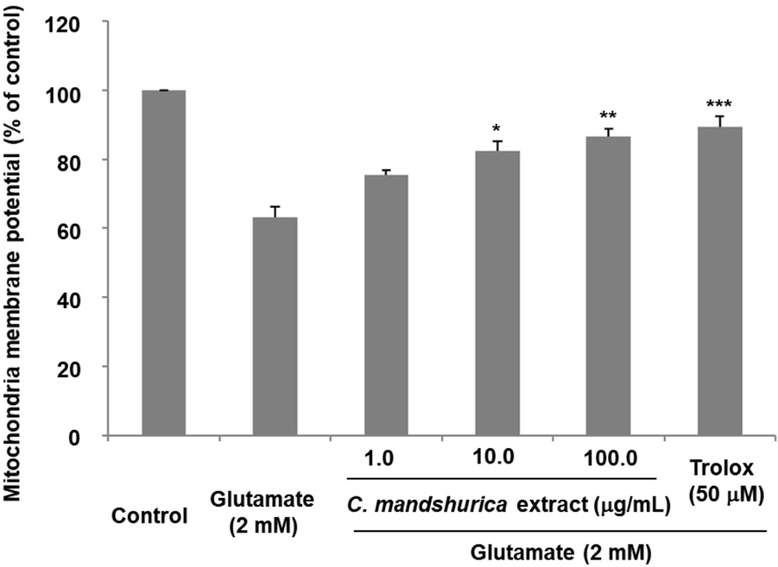
The effects of C. mandshurica extract on glutamate-induced loss of mitochondrial membrane potential in HT22 cells. HT22 cells were treated with C. mandshurica extract prior to glutamate exposure, and mitochondrial membrane potential was assessed using Rho123 staining. Results are expressed as a percentage relative to control cells. Data represent the mean ± SD of three independent experiments. *p < 0.05, **p < 0.01, ***p < 0.001 compared with glutamate-treated cells.
The glutathione system is one of the most important intracellular antioxidant defenses in neuronal cells. Glutamate-induced oxidative stress impairs glutathione synthesis by inhibiting cysteine uptake and decreases the activity of glutathione reductase (GR) and glutathione peroxidase (GPx).23) In accordance with this, glutamate treatment significantly depleted GSH (56.23 ± 2.3%), GR (54.58 ± 2.73%), and GPx (54.23 ± 3.45%) levels in HT22 cells. Remarkably, C. mandshurica pretreatment recovered these antioxidant markers. At 100 μg/ml, the extract restored GSH to 79.31 ± 1.5%, GR to 86.23 ± 3.27%, and GPx to 87.54 ± 3.69% of control levels (Fig. 5). These results demonstrate that C. mandshurica enhances endogenous antioxidant defense systems, thereby counteracting oxidative stress and reducing susceptibility to glutamate-induced neuronal death. Restoration of GSH and related enzyme activity is critical because GSH depletion leads to irreversible oxidative damage and mitochondrial dysfunction.24) Thus, the antioxidative capacity of C. mandshurica appears to be a major mechanism underlying its neuroprotective efficacy.
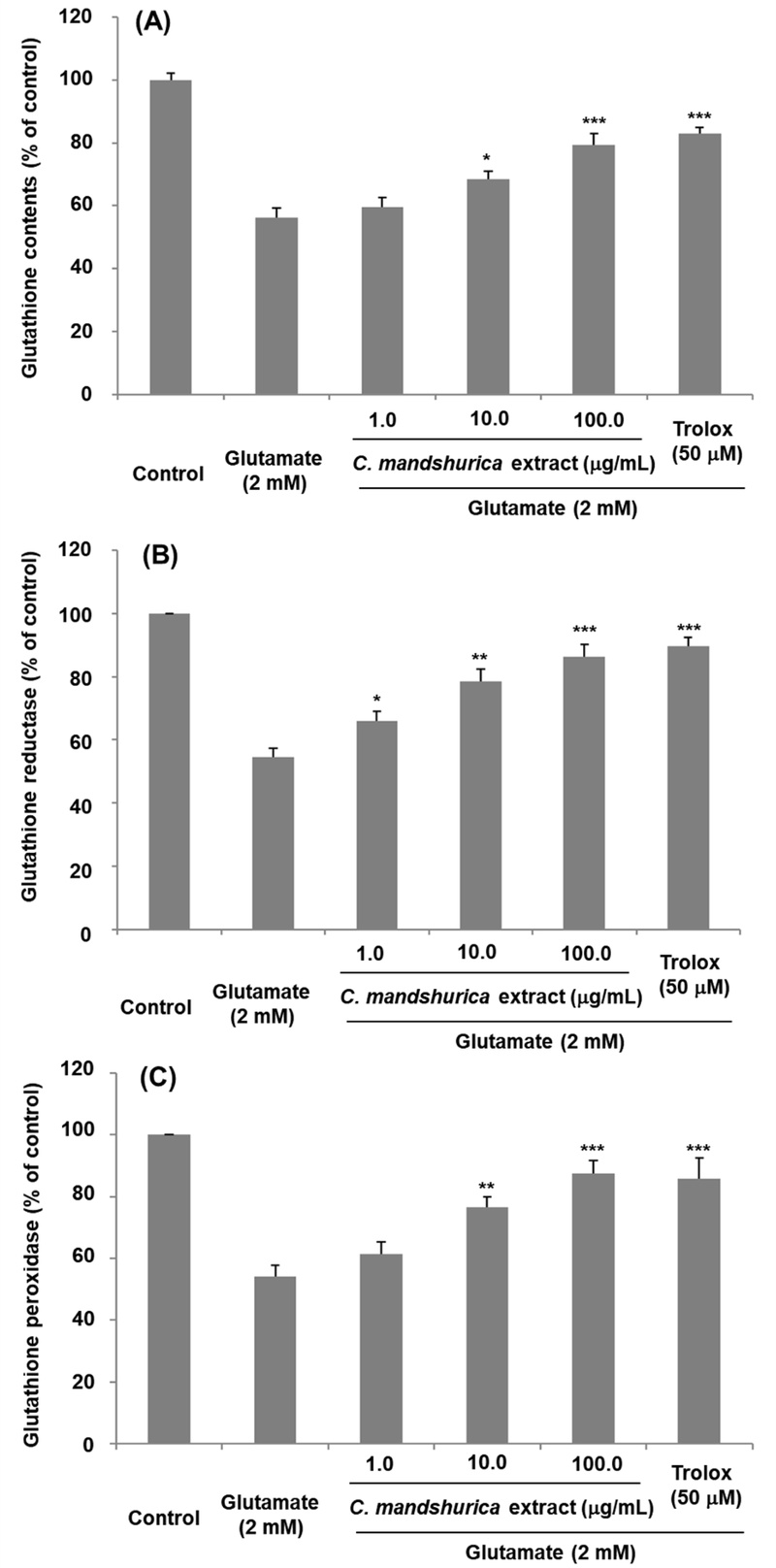
Effects of C. mandshurica extract on total glutathione (A), glutathione reductase (B), and glutathione peroxidase (C) levels in HT22 cells exposed to glutamate. HT22 cells were treated with C. mandshurica extract prior to glutamate exposure, and intracellular antioxidant enzyme activities (total GSH, GR, and GPx) were quantified. Results are expressed as percentages relative to control cells. Data represent the mean ± SD of three independent experiments. *p < 0.05, **p < 0.01, ***p < 0.001 compared with glutamate-treated cells.
The robust neuroprotective effect of C. mandshurica extract observed in this study can be attributed to its rich profile of bioactive secondary metabolites, particularly triterpenoid saponins and lignans.25) Previous phytochemical investigations have identified clematoside derivatives, such as clematoside C, as the hallmark triterpenoid saponins of the Clematis genus.26) These saponins are known to exhibit potent antiapoptotic properties by stabilizing the mitochondrial membrane potential and suppressing the overproduction of reactive oxygen species (ROS).27) Specifically, clematosides have been reported to upregulate endogenous antioxidant enzymes, which aligns with our findings regarding the recovery of glutathione (GSH), glutathione reductase (GR), and glutathione peroxidase (GPx) levels. Furthermore, the presence of lignans, such as lariciresinol and pinoresinol, likely contributes to the attenuation of Ca2+ influx.28) Lignans are well-documented for their ability to modulate calcium homeostasis and inhibit glutamate-induced excitotoxicity by interfering with overactive signaling pathways.29) These compounds may act synergistically with saponins to reinforce the neuronal defense system against oxidative stress and mitochondrial collapse. Therefore, the multi-targeted mechanism of C. mandshurica demonstrated in HT22 cells is likely a collective result of these specific chemical constituents.
In conclusion, we demonstrated that C. mandshurica extract exerts a robust neuroprotective effect against glutamate-induced cytotoxicity in HT22 neuronal cells. The extract significantly improved cell viability and effectively suppressed intracellular ROS production, indicating strong antioxidant activity. In addition, C. mandshurica attenuated glutamate-mediated Ca2+ influx and restored mitochondrial membrane potential, suggesting that its protective effects extend to the maintenance of calcium homeostasis and mitochondrial integrity. The extract also recovered glutamate-induced depletion of key antioxidant components, including total glutathione, glutathione reductase, and glutathione peroxidase. Collectively, these findings indicate that C. mandshurica mitigates glutamate-induced neuronal damage through multi-targeted mechanisms involving oxidative stress reduction, mitochondrial stabilization, and reinforcement of endogenous antioxidant defenses. Thus, C. mandshurica represents a promising natural candidate for the prevention or treatment of neurodegenerative disorders associated with glutamate excitotoxicity. However, further studies including in vivo validation, behavioral assessments, and detailed mechanistic analyses, will be essential to fully substantiate the therapeutic potential of C. mandshurica in neurodegenerative diseases.
Acknowledgments
This research was supported by Korea Basice Science Institute (National research Facilities and Equipment Center) grant funded by the Ministry of Education (grant No. 2022R1A6C101A739).
Conflict of Interest Statement
The authors have declared that there are no conflicts of interest.
References
-
DeTure, M. A. and Dickson, D. W. (2019) The neuropathological diagnosis of Alzheimer’s disease. Mol. Neurodegen. 14: 32.
[https://doi.org/10.1186/s13024-019-0333-5]

- Bertram, L. and Tanzi, R. E. (2012) The genetics of Alzheimer’s disease. Nat. Rev. Neurol. 8: 697-709.
-
Selkoe, D. J. and Hardy, J. (2016) The amyloid hypothesis of Alzheimer's disease at 25 years. EMBO Mol. Med. 8: 595-608.
[https://doi.org/10.15252/emmm.201606210]

-
McGeer, P. L. and McGeer, E. G. (2013) The amyloid cascade-inflammatory hypothesis of Alzheimer disease: implications for therapy. J. Neural Transm. 120: 519-526.
[https://doi.org/10.1007/s00401-013-1177-7]

-
Butterfield, D. A. and Halliwell, B. (2019) Oxidative stress, dysfunctional glucose metabolism and Alzheimer disease. Trends Neurosci. 42: 105-114.
[https://doi.org/10.1038/s41583-019-0132-6]

- Papadia, S. and Hardingham, G. E. (2007) The survival-promoting functions of extrasynaptic NMDA receptors in neurons. Biochim. Biophys. Acta 1772: 293-308.
- Kandiah, N., Chander, R. J., Ng, A., Narasimhalu, K., Lim, L., Zhang, A. and Lin, X. (2017) Cognitive decline in Alzheimer’s disease: A review of recent data. Ther. Adv. Neurol. Disord. 10: 113-121.
- Kennedy, D. O. and Scholey, A. B. (2006) The psycho-pharmacology of European herbs with cognition-enhancing properties. Phytother. Res. 20: 683-690.
-
Chang, B., Jin, C., Zhang, W., Kong, L., Yang, J. H., Lian, F. M., Li, Q. F., Yu, B., Liu, W. K., Yang, L. L., Zhao, P. and Zhen, Z. (2012) Antidiabetic effect of Clematis mandshurica Rupr. in diabetic mice. Am. J. Chin. Med. 40: 1177-1187.
[https://doi.org/10.1142/S0192415X12500875]

-
Yoon, W. J., Min, H. J., Cho, H. D., Kim, H. G., Park, W. L., Kim, D. H., Tachibana, H. and Seo, K. I. (2024) Bioactivity evaluation and phytochemical characterization of Euonymus alatus (Thunb.) Siebold leaf extract. Biomedicines 12: 2928.
[https://doi.org/10.3390/biomedicines12122928]

-
Kitanaka, S., Takido, M., Mizoue, K. and Nakaike, S. (1996) Triterpene saponins from Clematis chinensis Osbeck. Chem. Pharm. Bull. 44: 615-617.
[https://doi.org/10.1248/cpb.44.615]

- Li, X., Koike, K. and Ohmoto, T. (1993) Triterpenoid saponins from Clematis mandshurica Rupr. Chem. Pharm. Bull. 41: 1276-1278.
- Kim, C. H. (2003) A review on the pharmacological effects and clinical application of Clematidis Radix. Kor. J. Orient. Gynecol. 8: 28-36.
- Tan, S., Schubert, D. and Maher, P. (2001) Oxytosis: A novel form of programmed cell death. J. Neurochem. 78: 546-559.
-
Luo, Y. and DeFranco, D. B. (2006) Opposing roles for ERK1/2 in neuronal oxidative stress. J. Biol. Chem. 281: 624-633.
[https://doi.org/10.1074/jbc.M512430200]

- Maher, P. and Davis, J. B. (1996) The role of glutathione in neuronal survival and death. Brain Res. 706: 329-332.
-
Uttara, B., Singh, A. V., Zamboni, P. and Mahajan, R. T. (2009) Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr. Neuropharmacol. 7: 65-74.
[https://doi.org/10.2174/157015909787602823]

-
Dong, X., Wang, Y. and Qin, Z. (2009) Molecular mechanisms of excitotoxicity and their relevance to pathogenesis of neurodegenerative diseases. Acta Pharmacol. Sin. 30: 379-387.
[https://doi.org/10.1038/aps.2009.24]

-
Szydlowska, K. and Tymianski, M. (2010) Calcium, excitotoxicity and NMDA receptors: Role of calcium overload and signaling. Cell Calcium 47: 122-129.
[https://doi.org/10.1016/j.ceca.2010.01.003]

-
Abramov, A. Y., Scorziello, A. and Duchen, M. R. (2007) Three distinct mechanisms generate reactive oxygen species in neurons in response to glutamate. J. Neurosci. 27: 1129-1136.
[https://doi.org/10.1523/JNEUROSCI.4468-06.2007]

-
Nicholls, D. G. and Budd, S. L. (2000) Mitochondria and neuronal survival. Physiol. Rev. 80: 315-360.
[https://doi.org/10.1152/physrev.2000.80.1.315]

- Wang, X., Levi, M. S., Coyle, J. T., Feinstein, D. L. and Chen, S. (2004) Mitochondrial dysfunction and oxidative stress in neurodegeneration. J. Neurosci. Res. 76: 563-570.
-
Dringen, R., Gutterer, J. M. and Hirrlinger, J. (2000) Glutathione metabolism in brain: Gastrocytes and neurons. Eur. J. Biochem. 267: 4912-4916.
[https://doi.org/10.1046/j.1432-1327.2000.01597.x]

- Pérez-Severiano, F., Ríos, C. and Segovia, J. (2004) Striatal oxidative damage and mitochondrial dysfunction in a rat model of Huntington's disease. Neurochem. Int. 45: 471-478.
- Liu, M., Hong, B., Li, L., Wang, S., Chen, J. and Xu, Y. (2021) Phytochemical and pharmacological progress on the genus Clematis. Chin. J. Nat. Med. 19: 321-337.
- Li, F., Yan, X. P., Song, J. J., Tang, J. J., Ma, J. P. and Sun, J. P. (2018) Triterpenoid saponins from Clematis mandshurica and their protective effects against oxidative stress. J. Nat. Prod. 81: 920-928.
- Kim, S. Y., Park, S. J., Lee, K. W., Kim, J. H. and Lee, H. J. (2019) Clematoside C, a triterpenoid saponin, attenuates neuronal apoptosis by inhibiting mitochondrial dysfunction. Biol. Pharm. Bull. 42: 1011-1018.
- Zhang, Y., Liu, J., Chen, X., Wang, H., Zhao, L. and Zhang, S. (2020) Neuroprotective effects of lignans from the genus Clematis on glutamate-induced toxicity in hippocampal neurons. Phytomedicine 72: 153241.
- Wang, J. H., Luan, F., He, X. D., Wang, Y. and Li, M. X. (2015) Antioxidant and anti-inflammatory activities of extracts from Clematis mandshurica Rupr. J. Ethnopharmacol. 162: 242-249.