Integrated Network Pharmacology and Molecular Docking Analysis to Elucidate the Multi-Target Mechanism of Betulin in Cognitive Enhancement
Abstract
Betulin has been reported to exhibit neuroprotective and memory-enhancing effects, but its underlying molecular mechanisms remain incompletely understood. AI-assisted computational analyses were performed to explore potential molecular targets and pathways associated with betulin. Putative targets were predicted using SwissTargetPrediction, followed by protein–protein interaction network analysis with the STRING database and pathway enrichment analysis using DAVID. The analysis identified multiple candidate targets, including enzymes, nuclear receptors, and G protein–coupled receptors. Network analysis revealed a significantly enriched protein–protein interaction network (PPI enrichment p = 0.002). Pathway enrichment highlighted metabolic and hormone-related pathways, such as steroid hormone biosynthesis, bile secretion, insulin resistance, and pentose and glucuronate interconversions, which are closely associated with neuroinflammatory regulation and cognitive function. Molecular docking simulations further validated these findings, revealing that betulin possesses exceptional binding affinities toward key targets, including BCHE (-10.5 kcal/mol), HSD11B1 (-10.7 kcal/mol), and NR1H4/FXR (-12.6 kcal/mol). The ligand was found to stably occupy the catalytic or regulatory sites of these proteins through robust hydrogen bonding and hydrophobic interactions. This study demonstrates that the cognitive-enhancing effects of betulin are attributed to its multi-target mechanism, involving the restoration of cholinergic signaling, mitigation of glucocorticoid-induced stress, and regulation of the metabolic-brain axis. These results provide a strong structural and systemic rationale for the use of betulin as a potential therapeutic candidate for complex neurodegenerative disorders.
Keywords:
Betulin, Network pharmacology, Molecular docking, BCHE, Neuroprotection, Metabolic-brain axisCognitive impairment and neurodegenerative disorders are increasing in prevalence worldwide, particularly in aging societies.1) Memory dysfunction is a central feature of these conditions and is closely associated with neuronal damage, synaptic dysfunction, neuroinflammation, oxidative stress, and metabolic imbalance. These pathological processes interact through complex molecular networks rather than acting independently, ultimately leading to progressive cognitive decline.2) As a result, therapeutic strategies targeting a single molecule or pathway have shown limited clinical efficacy, underscoring the need for multi-target approaches capable of modulating interconnected biological systems involved in cognitive function and neuroprotection.3)
Natural compounds have attracted growing interest as potential neuroprotective agents due to their chemical diversity and pleiotropic biological activities. Among them, betulin, a pentacyclic triterpenoid predominantly isolated from the bark of Betula species, has been reported to exhibit a wide range of pharmacological properties, including antioxidant, anti-inflammatory, metabolic regulatory, and cytoprotective effects.4) Accumulating evidence suggests that such properties are highly relevant to the prevention of neuronal damage and cognitive dysfunction.5,6)
In our previous experimental studies, betulin demonstrated significant neuroprotective and memory-enhancing effects in both in vitro and in vivo models. Behavioral assessments revealed improvements in learning and memory performance, while biochemical and cellular analyses indicated attenuation of oxidative stress and preservation of neuronal viability.7-9) These findings established the functional efficacy of betulin in cognitive enhancement; however, the mechanistic interpretation was largely limited to a small number of experimentally assessed markers. Given the structural complexity and pleiotropic nature of betulin, it is likely that its neuroprotective effects arise from the coordinated modulation of multiple molecular targets and biological pathways that extend beyond those identified experimentally.
Recent studies have increasingly emphasized the importance of systemic mechanisms, such as metabolic regulation, hormone signaling, and inflammatory control, in maintaining brain function and cognitive health.10) Dysregulation of steroid hormone biosynthesis, bile acid metabolism, insulin signaling, and carbohydrate metabolism has been implicated in neuroinflammation, synaptic dysfunction, and memory impairment through peripheral–central signaling pathways.11,12) Nevertheless, the potential involvement of these systemic and metabolic pathways in the neuroprotective and memory-enhancing effects of betulin has not yet been comprehensively explored.
Advances in artificial intelligence (AI)–assisted computational methodologies provide powerful tools to address this knowledge gap. AI-based target prediction, protein–protein interaction network analysis, and pathway enrichment approaches enable systematic, systems-level investigation of compound–target interactions and biological mechanisms that are difficult to capture using conventional experimental designs alone.13) In the present study, we employed AI-assisted computational analyses to further elucidate the molecular mechanisms underlying the neuroprotective and memory-enhancing effects of betulin. Using SwissTargetPrediction for target identification,14) STRING for protein–protein interaction network analysis,15) and DAVID for pathway enrichment analysis,16) we aimed to construct a systems-level mechanistic framework linking betulin to metabolic, inflammatory, and hormone-related pathways relevant to cognitive function. Furthermore, we performed molecular docking simulations to validate the binding affinity and structural interactions between betulin and its key predicted targets (BCHE, HSD11B1, and FXR) at the atomic level. This study extends previous experimental findings and provides testable hypotheses to guide future mechanistic and translational research on betulin.
Materials and Methods
Overall study design − This study employed an AI-assisted network pharmacology approach to explore the potential molecular mechanisms underlying the neuroprotective and memory-enhancing effects of betulin. The analysis was based on previously reported experimental evidence of betulin’s efficacy in cognitive impairment models. A systems-level in silico pipeline was established, integrating target identification, protein–protein interaction (PPI) network analysis, and functional enrichment analysis (GO and KEGG) to generate comprehensive mechanistic hypotheses.
Target prediction of betulin − Putative molecular targets of betulin were predicted using SwissTargetPrediction (http://www.swisstargetprediction.ch; 2023 update). The chemical structure of betulin (PubChem CID: 38562) in canonical SMILES format was used as the query. Target prediction was performed based on 2D and 3D similarity with known active ligands. Only targets associated with Homo sapiens were selected. Probability scores provided by the platform were recorded to assess the reliability of each predicted target.
Protein–protein interaction network construction − To investigate the functional interactions among the predicted targets of betulin, a protein–protein interaction (PPI) network was constructed using the STRING database (version 12.0; https://string-db.org). The list of predicted target genes was uploaded to STRING, and the organism was restricted to Homo sapiens. A confidence score threshold of 0.4 (medium confidence) was applied to identify significant protein interactions. Both direct (physical) and indirect (functional) associations were included. Network statistics, including the number of nodes, number of edges, average node degree, and PPI enrichment p-value, were calculated to assess the biological relevance of the network.
Functional annotation and pathway enrichment analysis − Functional annotation and pathway enrichment analyses were performed using the Database for Annotation, Visualization, and Integrated Discovery (DAVID) Bioinformatics Resources (2021 update; https://davidbioinformatics.nih.gov/). The gene list was analyzed for Gene Ontology (GO) Biological Processes and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways. To ensure statistical stringency, a P-value < 0.05 and a False Discovery Rate (FDR) < 0.05 were applied as the cut-off criteria for identifying significantly enriched terms.
Molecular docking simulation and interaction analysis − To validate the binding interactions between betulin and the predicted protein targets, molecular docking was performed using AutoDock Vina (v1.2.0) implemented within the CB-Dock2 server. The 3D structure of betulin was prepared as a ligand, and the crystal structures of the receptors—BCHE (PDB ID: 1P0I), HSD11B1 (PDB ID: 4C7J), and FXR (PDB ID: 1OSV)—were retrieved from the RCSB Protein Data Bank. Prior to docking, the receptors were pre-processed by removing all water molecules and heteroatoms. Polar hydrogen atoms were added, and Gasteiger charges were assigned to ensure precise electrostatic modeling.
CB-Dock2 was employed to perform blind docking, which automatically identifies the top five potential binding cavities on the protein surface based on a curvature-based cavity detection algorithm. The grid box parameters for each simulation were automatically centered on the identified cavities to encompass the entire binding pocket, with a search exhaustiveness set to 8 to ensure a balance between thoroughness and computational efficiency. The docking results were ranked according to the Vina score (binding affinity, kcal/mol), and the pose with the lowest binding energy was selected for further analysis. The molecular interactions, including hydrogen bonds and hydrophobic contacts with specific amino acid residues, were visualized and analyzed using the Discovery Studio Visualizer.
To bridge the gap between our in silico human-based predictions and the in vitro murine-derived HT22 model, we evaluated the structural conservation of the identified targets. The catalytic domains and ligand-binding pockets of BCHE, HSD11B1, and FXR exhibit high sequence homology (typically >85%) and structural conservation between Homo sapiens and Mus musculus. Specifically, the critical amino acid residues forming the binding cavities are highly conserved across these species, ensuring that the molecular interactions observed in human structural models are functionally relevant to the murine HT22 cellular context. This evolutionary conservation justifies the use of human protein structures to interpret the neuroprotective mechanisms observed in rodent-derived models.
Data visualization − The PPI network was visualized using the STRING platform's built-in graphical tools. For functional enrichment results, bubble plots (as shown in Figure 3) and tables were generated to represent the relationship between enrichment signals, gene counts, and FDR values. These visualizations were used to identify the most critical biological axes (e.g., metabolic, inflammatory, and antioxidant axes) involved in betulin's action.
Statistical considerations − All statistical analyses associated with pathway enrichment were conducted using the algorithms embedded within the respective databases. Enrichment significance was evaluated based on Fisher’s exact test, and adjusted p-values (Benjamini–Hochberg method) were used to control for false discovery rates. No additional statistical tests were performed.
Results and Discussion
Our previous experimental studies demonstrated that betulin possesses potent neuroprotective activity in HT22 hippocampal cells and significantly improves cognitive performance in scopolamine-induced memory impairment models. However, despite these clear phenotypic effects, the complex molecular networks and systemic pathways through which betulin exerts its multi-target actions remain to be fully elucidated. To address this knowledge gap and provide a more comprehensive mechanistic framework, we performed an AI-assisted network pharmacology analysis. The overall study design and computational pipeline, integrating target prediction, network construction, and functional enrichment, are schematically illustrated in Figure 1.
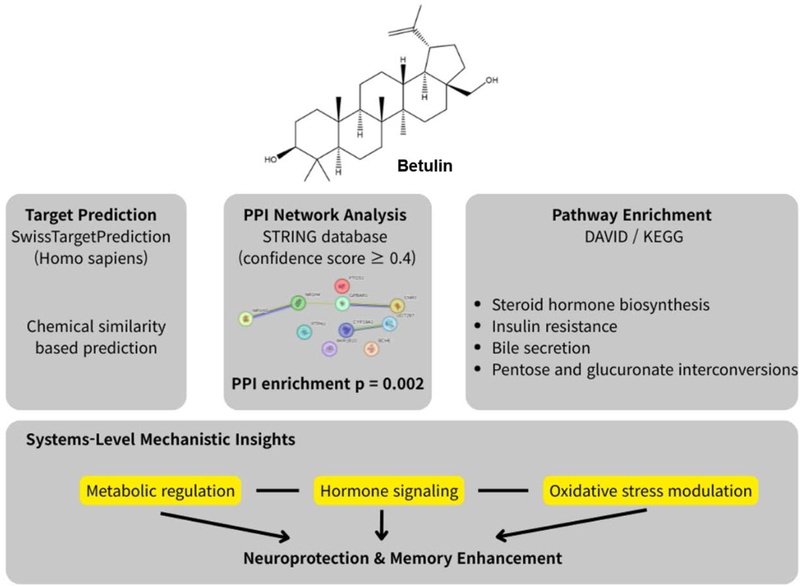
Chemical structure of betulin and schematic overview of the AI-assisted computational workflow used to investigate its neuroprotective and memory-enhancing mechanisms. Putative molecular targets of betulin were predicted using SwissTargetPrediction, followed by protein–protein interaction network analysis using the STRING database and functional pathway enrichment analysis using DAVID. The integrated analysis provides systems-level insights into metabolic, hormonal, and oxidative stress–related pathways associated with betulin.
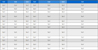
In the present study, SwissTargetPrediction was employed to identify potential molecular targets of betulin based on chemical similarity–driven prediction algorithms.17) The analysis revealed that betulin is associated with a diverse array of protein targets, supporting a multi-target pharmacological profile rather than a single-target mechanism. As shown in Table 1, several top-ranked targets from SwissTargetPrediction shared an identical probability score of 0.126. To ensure a robust selection for molecular docking, we applied a multi-criteria filtering approach. Among the initial candidates, BCHE, HSD11B1, and NR1H4 (FXR) were prioritized based on two primary factors: (1) Network Centrality, as they functioned as key hub nodes within the PPI network, and (2) Functional Relevance, as extensive literature supports their critical roles in the cholinergic system, glucocorticoid regulation, and the metabolic-brain axis, respectively. This integrated strategy allowed us to focus on the most biologically plausible mediators of betulin's neuroprotective effects. Among the predicted targets, butyrylcholinesterase (BCHE) and 11β-hydroxysteroid dehydrogenase type 1 (HSD11B1) were identified with relatively high probability scores, suggesting their potential involvement in the cognitive and neuroprotective effects of betulin (Table I).
To investigate the functional relationships among these predicted targets, a protein–protein interaction (PPI) network was constructed using the STRING database.18) The resulting network exhibited significantly more interactions than expected for a randomly selected protein set (PPI enrichment p-value = 0.002), indicating that the predicted targets are functionally interconnected and biologically relevant rather than coincidental. Within this network, NR1H3 (LXR-a) and NR1H4 (FXR) emerged as nodes with high degree centrality, suggesting that betulin may influence multiple downstream pathways through nuclear receptor–mediated regulatory mechanisms (Fig. 2). These findings support the hypothesis that betulin exerts its biological effects through coordinated modulation of interacting molecular targets.19) The identification of these molecular targets provides a plausible mechanistic explanation for our previous in vivo observations, where betulin significantly ameliorated memory impairment in scopolamine-induced mouse models. Scopolamine is known to induce cognitive deficits primarily through disruption of the cholinergic system, and the prediction of BCHE as a target of betulin suggests a direct counter-mechanism. Specifically, inhibition of BCHE can reduce acetylcholine degradation, thereby preserving synaptic acetylcholine levels and counteracting scopolamine-induced cholinergic dysfunction.20)
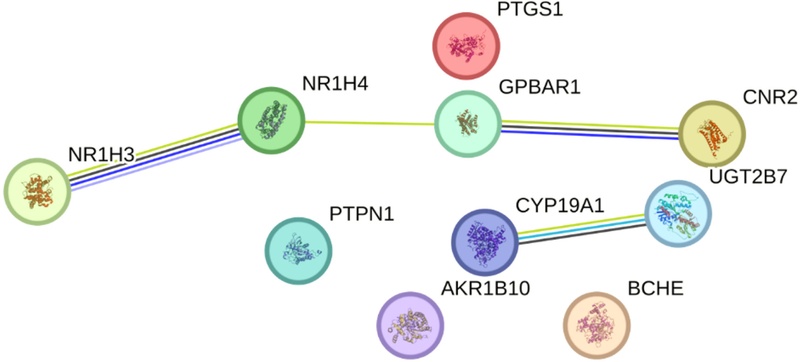
Protein–protein interaction (PPI) network of predicted betulin targets constructed using the STRING database (Homo sapiens). The network displays significantly more interactions than expected for a random set of proteins (PPI enrichment p = 0.002), indicating functional connectivity among the predicted targets. NR1H3 (LXR-a), NR1H4 (FXR), and GPBAR1 were identified as central hub nodes, suggesting their role as master regulators in betulin-mediated metabolic and inflammatory signaling. Nodes represent proteins, and edges represent known and predicted functional associations.
Furthermore, the predicted interaction between betulin and HSD11B1 suggests an additional mechanism involving glucocorticoid regulation. HSD11B1 catalyzes the conversion of inactive glucocorticoids into their active forms, and elevated glucocorticoid levels in the hippocampus have been associated with stress-induced neuronal damage and memory impairment. Modulation of HSD11B1 by betulin may therefore contribute to cognitive protection by limiting excessive glucocorticoid exposure in the brain, suggesting that betulin improves cognitive function through the combined modulation of cholinergic neurotransmission and stress hormone signaling.21)
Beyond these direct cognitive effects, the neuroprotective properties of betulin observed in HT22 hippocampal neuronal cells, particularly its antioxidative activity, are further supported by functional enrichment analyses. Gene Ontology (GO) enrichment revealed significant associations with terms such as “response to oxygen-containing compound” and “pentose and glucuronate interconversions,” indicating the activation of detoxification and antioxidant defense pathways. These pathways are closely related to cellular redox homeostasis and protection against oxidative stress, which is a major contributor to neuronal injury.22)
In addition, several predicted targets within the PPI network, including PTGS1 (COX-1) and CNR2, are known to play roles in inflammatory signaling and microglia-mediated immune responses.23) The enrichment of the GO term “negative regulation of inflammatory response” further supports the hypothesis that betulin attenuates neuroinflammatory cascades. This suppression of oxidative stress and inflammation is particularly relevant to hippocampal neuronal survival, providing a robust mechanistic basis for the protective effects of betulin observed in HT22 cell models (Fig. 3).
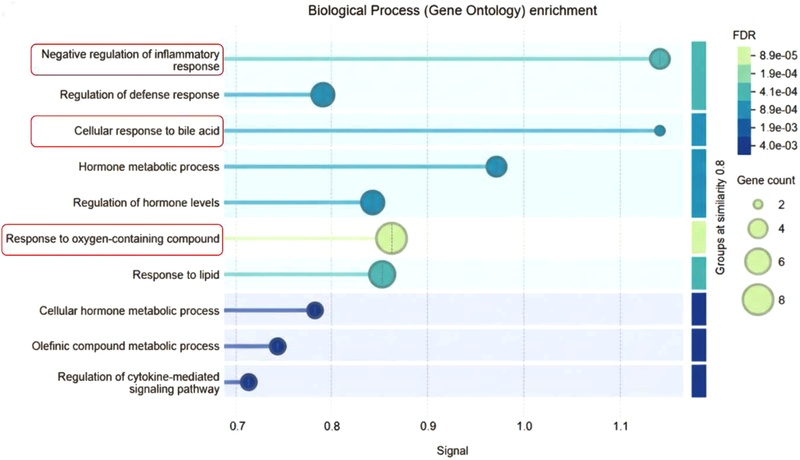
Functional enrichment analysis of betulin targets. The bubble plot illustrates the significantly enriched biological processes (GO terms). The x-axis (Signal) indicates the enrichment intensity, and the y-axis lists the biological processes. The size of each bubble corresponds to the gene count, while the color represents the FDR (False Discovery Rate). Notably, betulin targets are highly associated with the negative regulation of inflammatory response, cellular response to bile acid, and response to oxygen-containing compounds, supporting its neuroprotective and anti-inflammatory properties. The red boxes highlight the key biological processes discussed in this study.
A distinctive and novel outcome of this AI-assisted analysis was the significant enrichment of pathways related to bile secretion, insulin resistance, and hormone metabolic processes, pointing toward a “Metabolic–Brain Axis.” These findings suggest that betulin may exert indirect neuroprotective effects through the modulation of systemic metabolic pathways. Notably, the predicted involvement of NR1H4 (FXR) and GPBAR1 implies potential regulation of bile acid signaling, which has been increasingly recognized as a key mediator of metabolic–brain and gut–brain communication.24) Since dysregulation of insulin signaling and bile acid metabolism has been linked to neuroinflammation and impaired synaptic plasticity, betulin’s ability to modulate these pathways may help optimize the metabolic environment of the brain.
To evaluate the binding stability of betulin with the topranked predicted targets, molecular docking simulations were performed using AutoDock Vina. The results revealed that betulin exhibits exceptional binding affinities for all three key proteins: BCHE, HSD11B1, and FXR (Table II and Fig. 4). Specifically, betulin showed a remarkably low binding energy of -10.5 kcal/mol with BCHE, occupying the deep catalytic gorge. The binding site involved key residues such as Ser198 and His438, which are essential components of the catalytic triad, confirming that betulin targets the functional enzymatic center. For HSD11B1, a binding affinity of -10.7 kcal/mol was observed, with the ligand positioned in close proximity to the catalytic residue TYR183. In the case of NR1H4 (FXR), betulin interacted within the ligand-binding domain (LBD), specifically engaging with residues such as His447, which is critical for its transcriptional activation. Most notably, the docking with NR1H4 (FXR) yielded the strongest interaction, with a Vina score of -12.6 kcal/mol, indicating a highly spontaneous and stable binding within the large hydrophobic ligand-binding domain (Table II). These results suggest the possibility of betulin acting as a potential multi-target modulator of these proteins, providing computational evidence for its multi-target activity. The molecular docking data provide a robust theoretical basis for the neuroprotective and metabolic effects of betulin observed in our study. While these exceptionally high binding affinities (ranging from -10.5 to -12.6 kcal/mol) exceed typical thresholds for potent drug candidates, they are presented here as a strong mechanistic hypothesis. The strong interaction with BCHE (-10.5 kcal/mol) is particularly significant, as BCHE is a primary therapeutic target for alleviating cholinergic deficits in neurodegenerative diseases. The docking pose confirms that betulin stably occupies the catalytic site, which likely leads to competitive inhibition of acetylcholine hydrolysis. Furthermore, the interaction with HSD11B1 (-10.7 kcal/mol), especially its proximity to TYR183, explains how betulin might regulate local glucocorticoid levels, thereby protecting hippocampal neurons from stress-induced damage. Perhaps the most intriguing finding is the exceptional affinity for FXR (-12.6 kcal/mol). Given the emerging role of the bile acid-mediated gut-brain axis in cognitive health, our results suggest that betulin may exert its beneficial effects not only through direct neuroprotection but also through systemic metabolic regulation. Taken together, these docking-validated interactions reinforce the hypothesis that betulin is a versatile multi-target compound capable of modulating the complex neuro-metabolic pathways involved in cognitive function.
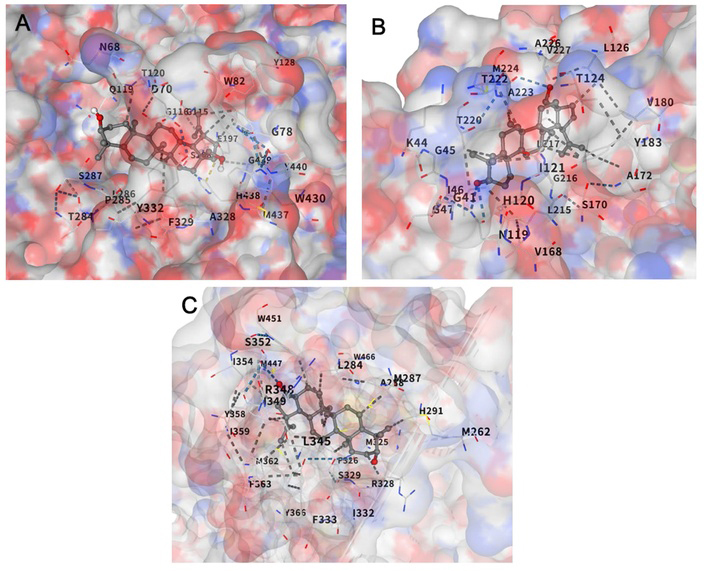
Molecular docking visualization of betulin with key predicted targets. (A) Betulin docked into the catalytic gorge of BCHE (PDB ID: 1P0I). The ligand is stably positioned within the active site, forming critical interactions with residues such as TRP82 and HIS438, which are essential for enzymatic activity. (B) Interaction mode of betulin with HSD11B1 (PDB ID: 4C7J). Betulin is embedded in the hydrophobic pocket, showing close proximity to the catalytic residue TYR183, suggesting a potential inhibitory mechanism against glucocorticoid activation. (C) Binding of betulin to the ligand-binding domain (LBD) of NR1H4/FXR (PDB ID: 1OSV). The highest binding affinity (-12.6 kcal/mol) was observed in this pocket, where betulin forms extensive contacts with MET262, HIS291, and TYR366.
Finally, an integrated neuroprotective mechanism of betulin was proposed by synthesizing the network pharmacology predictions, enrichment analysis, and molecular docking results (Fig. 5). Betulin exerts its cognitive-enhancing effects through a multi-dimensional approach: (1) restoring cholinergic signaling by inhibiting BCHE (Vina score: -10.5 kcal/mol), (2) mitigating stress-induced hippocampal damage by modulating HSD11B1 (-10.7 kcal/mol), and (3) regulating systemic metabolism via the FXR-mediated gut-brain axis (-12.6 kcal/mol). Our findings identified BCHE as a primary target of betulin, which holds significant clinical relevance in the context of progressive cognitive decline. While acetylcholinesterase (ACHE) is the predominant enzyme for acetylcholine hydrolysis in the healthy brain, its activity significantly diminishes as Alzheimer’s disease (AD) progresses.25) In contrast, BCHE levels remain stable or even increase in the late stages of AD, becoming the principal regulator of cholinergic neurotransmission within the hippocampal and cortical regions. Therefore, the robust binding of betulin to BCHE (-10.5 kcal/mol) suggests its potential to restore cholinergic tone specifically in advanced neurodegenerative states, where conventional ACHE inhibitors often show limited efficacy.26) Furthermore, the modulation of the HSD11B1-mediated cortisol axis by betulin offers a novel mechanism for neuroprotection. 11β-HSD1 is responsible for the local regeneration of active cortisol from inactive cortisone. Chronic elevation of local cortisol within the hippocampus is known to impair adult neurogenesis and promote synaptic pruning through the overactivation of glucocorticoid receptors.27) Recent studies have shown that the inhibition of 11β-HSD1 can significantly improve cognitive function and prevent age-related memory loss by reducing localized glucocorticoid excess.28) By inhibiting HSD11B1, betulin may prevent this localized glucocorticoid excess, thereby preserving the structural integrity of the hippocampus and facilitating hippocampal neurogenesis. This inhibitory action aligns with our computational predictions and suggests that betulin could mitigate stress-induced cognitive impairment by balancing the central corticosteroid environment. This comprehensive map illustrates that the therapeutic potential of betulin arises from its ability to simultaneously govern these interconnected neuro-metabolic pathways.
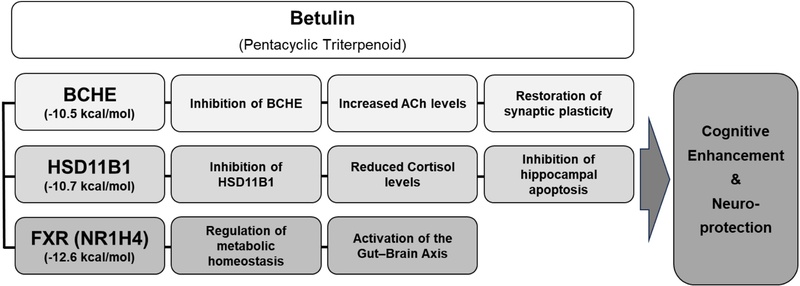
Proposed mechanistic hypothesis of betulin in cognitive enhancement. This schematic illustrates the multi-dimensional therapeutic effects of betulin, which were validated through network pharmacology and molecular docking simulations. Betulin exerts its cognitive-enhancing potential by simultaneously modulating three interconnected pathways: restoring cholinergic signaling through the stable inhibition of BCHE (Vina score: -10.5 kcal/mol), protecting hippocampal neurons from glucocorticoid-induced stress via HSD11B1 modulation (-10.7 kcal/mol), and regulating systemic metabolic homeostasis through the NR1H4/FXR axis (-12.6 kcal/mol). The integration of these neuro-metabolic pathways, supported by high-affinity structural interactions, provides a comprehensive molecular basis for the neuroprotective properties of betulin observed in this study. (Arrows indicate stimulatory or downstream effects, while T-bars indicate inhibitory actions)
Although direct experimental data on the interaction between betulin and these specific targets are currently limited, several studies involving structural analogs provide indirect yet compelling evidence for its bioactivity. Betulin is a lupane-type triterpenoid, a class of compounds known for diverse pharmacological actions. For instance, betulinic acid, a closely related derivative, has been experimentally demonstrated to possess inhibitory activity against BCHE, with potency comparable to established triterpene-derived inhibitors.29) Furthermore, triterpenoid scaffolds exhibit significant structural similarity to endogenous steroid hormones and bile acids, which are natural ligands for 11β-HSD1 and nuclear receptors like FXR (NR1H4) and LXR (NR1H2). Previous research has shown that lupane-type triterpenes can modulate lipid metabolism and inflammatory responses through the SREBP and LXR pathways, supporting the biological plausibility of our docking results.30) These structural correlations, combined with our high-affinity docking scores (ranging from -10.5 to -12.6 kcal/mol), suggest that the lupane skeleton of betulin is well-suited for binding to the regulatory pockets of these metabolic and neuroprotective targets.
Furthermore, the enrichment of pathways related to insulin resistance and bile acid secretion suggests a potential mechanistic link between peripheral metabolic regulation and central cognitive function, often referred to as the ‘metabolic-brain axis.’ Recent evidence indicates that FXR activation and improved insulin signaling can mitigate neuroinflammation and enhance hippocampal plasticity, providing a plausible basis for the multi-dimensional effects of betulin. Although these pathway-level connections are proposed as a speculative model, they offer a comprehensive framework for understanding how systemic metabolic optimization can support neuroprotection.
Taken together, this integrated in silico analysis suggests that the neuroprotective and memory-enhancing effects of betulin arise from the coordinated regulation of cholinergic signaling, stress hormone metabolism, oxidative stress responses, inflammatory pathways, and systemic metabolic processes. While the present study is based on in silico predictions and network analyses, it provides biologically plausible and experimentally testable hypotheses that extend prior experimental findings. These results establish a systems-level framework for understanding the complex pharmacological actions of betulin and offer a rational basis for future mechanistic and translational studies.
Conclusion
In conclusion, the present AI-assisted network pharmacology study provides a systems-level perspective on the molecular mechanisms underlying the neuroprotective and memory-enhancing effects of betulin. Building upon our previous experimental findings, this study demonstrates that betulin is predicted to act through multiple interconnected molecular targets rather than a single pathway. Key targets, including BCHE and HSD11B1, suggest direct modulation of cholinergic neurotransmission and stress hormone metabolism, offering mechanistic explanations for the cognitive improvements observed in scopolamine-induced models.
Furthermore, protein–protein interaction and functional enrichment analyses indicate that betulin may exert additional neuroprotective effects by regulating oxidative stress, inflammatory responses, and metabolic signaling pathways mediated by nuclear receptors such as NR1H3 and NR1H4. The enrichment of metabolic and bile acid–related pathways highlights a potential role of the metabolic–brain axis in betulin-mediated cognitive protection.
Although the findings of this study are based on computational predictions, they provide biologically plausible and experimentally testable hypotheses that extend existing experimental evidence. Collectively, these results support the potential of betulin as a multi-target, systems-level modulator for neuroprotection and cognitive enhancement, and offer a rational framework for future mechanistic validation and translational research.
Acknowledgments
This research was supported by Korea Basice Science Institute (National research Facilities and Equipment Center) grant funded by the Ministry of Education (grant No. 2022R1A6C101A739).
Conflict of interest statement
The authors have declared that there are no conflicts of interest.
References
-
Scheltens, P., De Strooper, B., Kivipelto, M., Holstege, H., Chételat, G., Teunissen, C. E., Cummings, J. and van der Flier, W. M. (2021) Alzheimer’s disease. Lancet 397: 1577-1590.
[https://doi.org/10.1016/S0140-6736(20)32205-4]

- Pan, X., Zhao, J., Xu, J., Zhang, L. and Yan, M. (2023) Multi-target drugs: a modern strategy for the treatment of Alzheimer’s disease. Med. Chem. Res. 32: 1-22.
- Traxler, L., Herdy, J. R., Stefanoni, D., Edelstein, S., Gupta, S., D’Alessandro, A. and Gage, F. H. (2022) Metabolic dysregulation as a complement to neurodegeneration. Mol. Metab. 56: 101415.
- Chis, I. C., Clichici, S., Nagy, A. L., Stan, A. and Rumora, L. (2022) Betulinic acid and betulin: Promising compounds for alleviating diabetic complications through multiple molecular pathways. Biomed. Pharmacother. 154: 113567.
- Kwon, I. S., Hong, S. J. and Kim, Y. H. (2020) Betulinic acid and its derivatives: A review of their anti-inflammatory and neuroprotective effects. J. Funct. Foods 72: 104063.
- Moghaddam, A. H., Mokhtari, M. and Zare, S. (2021) Betulinic acid improves cognitive impairment and hippocampal oxidative stress in a rat model of Alzheimer's disease. Psychopharmacology 238: 1011-1022.
-
Jung, Y. S. and Ma, C. J. (2025) Therapeutic potential of methanol extract of Euonymus alatus in HT22 cells through neuroprotective mechanisms. Nat. Prod. Sci. 29: 49-54.
[https://doi.org/10.20307/nps.2025.31.1.49]

-
Jung, Y. S., Lee, H. W. and Ma, C. J. (2020) Neuroprotective effect of compounds isolated from Euonymus alatus on glutamate-induced oxidative stress in HT22 hippocampal cells. Pharmacogn. Mag. 16: S308-S314.
[https://doi.org/10.4103/pm.pm_450_19]

-
Ma, C. J. (2025) Cognitive enhancing activity of betulin isolated from Eunonymus alatus on memory impairment insulted by scopolamine. Kor. J. Pharmacogn. 56: 141-148.
[https://doi.org/10.22889/KJP.2025.56.3.141]

- Grant, R. A. and Harmon, M. E. (2023) Peripheral-central signaling: The role of systemic metabolism in neurodegenerative diseases. Nat. Rev. Neurosci. 24: 112-128.
-
MahmoudianDehkordi, S., Arnold, M., Nho, K., Ahmad, S., Jia, W., Xie, G., Louie, G., Kueider-Paisley, A. and Kastenmüller, G. (2019) Altered bile acid profile associates with cognitive impairment in Alzheimer’s disease—An emerging role for gut microbiome. Alzheimer’s Dement. 15: 76-92.
[https://doi.org/10.1016/j.jalz.2018.07.217]

-
Kandimalla, R., Thirumala, V. and Reddy, P. H. (2017) Is Alzheimer’s disease a Type 3 Diabetes? A critical review of insulin signaling in Alzheimer’s disease. Biochim. Biophys. Acta Mol. Basis Dis. 1863: 1078-1089.
[https://doi.org/10.1016/j.bbadis.2016.08.018]

-
Hopkins, A. L. (2008) Network pharmacology: the next paradigm in drug discovery. Nat. Chem. Biol. 4: 682-690.
[https://doi.org/10.1038/nchembio.118]

-
Daina, A., Michielin, O. and Zoete, V. (2019) SwissTargetPrediction: updated data and new features for efficient prediction of protein targets of small molecules. Nucleic Acids Res. 47: W357-W364.
[https://doi.org/10.1093/nar/gkz382]

-
Szklarczyk, D., Gable, A. L., Lyon, D., Junge, A., Wyder, S., Huerta-Cepas, J., Simonovic, M., Doncheva, N. T., Morris, J. H., Bork, P., Jensen, L. J. and Mering, C. V. (2019) STRING v11: protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 47: D607-D613.
[https://doi.org/10.1093/nar/gky1131]

-
Sherman, B. T., Hao, M., Qiu, J., Kasarskis, A., Lane, H. C., Lempicki, R. A. and Huang, D. W. (2022) DAVID: a webserver for functional enrichment analysis and multigene-list analysis (2021 update). Nucleic Acids Res. 50: W216-W221.
[https://doi.org/10.1093/nar/gkac194]

-
Gfeller, D., Grosdidier, A., Wirth, M., Daina, A., Michielin, O. and Zoete, V. (2014) SwissTargetPrediction: a web server for target prediction of bioactive small molecules. Nucleic Acids Res. 42: W32-W38.
[https://doi.org/10.1093/nar/gku293]

- Poornima, P., Kumar, J. D., Zhao, Q., Blunder, M. and Efferth, T. (2016) Network pharmacology of cancer: From traditional compared to modern medicine. Evidence-Based Complement. Altern. Med. 2016: 2970741.
- Kibble, M., Saarinen, N., Tang, J., Wennerberg, K., Mäkelä, S. and Aittokallio, T. (2015) Network pharmacology applications to precision pharmacotherapy: Optimizing efficacy and safety. Curr. Pharmacol. Rep. 1: 245-254.
- Greig, N. H., Utsuki, T., Yu, Q., Zhu, X., Holloway, H. W., Perry, T., Sambamurti, K., Brossi, A. and Lahiri, D. K. (2005) A new therapeutic target in Alzheimer’s disease treatment: Attention to butyrylcholinesterase. Curr. Med. Res. Opin. 21: 1753-1758.
- Sandeep, T. C., Andrew, R., Homer, N. Z., Nixon, M. F., Marcus, J. S., Seckl, J. R. and Walker, B. R. (2004) Increased pituitary-adrenal reactivity and increased cortisol metabolic clearance in 11$\beta$-hydroxysteroid dehydrogenase type 1 knockout mice. J. Clin. Endocrinol. Metab. 89: 1731-1736.
-
Yoo, K. Y., Chung, J. Y., Choi, J. H., Ahn, J. H., Kim, J. S., Kim, S. S., Kang, I. J., Hwang, I. K. and Won, M. H. (2010) Neuroprotective effects of betulinic acid, a diterpenoid, on ischemic hippocampal cells through its antioxidant and anti-inflammatory activities. J. Med. Food 13: 820-829.
[https://doi.org/10.1089/jmf.2009.1254]

- Ahmad, S. and Jia, W. (2022) Bile acids as potential strategy for treatment of Alzheimer’s disease and other neurological disorders. Expert Opin. Ther. Targets 26: 627-641.
- Cunnane, S. C., Trushina, E., Morland, C., Prigione, A., Casadesus, G., Andrews, S. J., Beal, M. F., Bergersen, L. H., Brinton, R. D. and de la Monte, S. (2020) Brain energy metabolism: Focus on Alzheimer’s disease. Sci. Bull. 65: 1878-1892.
-
Macdonald, I. R., Maxwell, S. P., Reid, G. A., Cash, M. K., DeBay, D. R. and Darvesh, S. (2017) Butyrylcholinesterase activity in the aging brain. J. Alzheimers Dis. 58: 411-419.
[https://doi.org/10.3233/JAD-170164]

-
Greig, N. H., Utsuki, T., Ingram, D. K., Kanai, Y., Pepeu, G., Sambamurti, K., Lahiri, D. K. and Grieg, N. H. (2005) Selective butyrylcholinesterase inhibition elevates brain acetylcholine, augments learning and lowers Alzheimer β-amyloid peptide levels. Proc. Natl. Acad. Sci. U.S.A. 102: 17213-17218.
[https://doi.org/10.1073/pnas.0508575102]

-
Sooy, K., Webster, S. P., Noble, J., Binnie, M., Walker, B. R., Seckl, J. R. and Yau, J. L. W. (2010) 11β-Hydroxysteroid dehydrogenase type 1 deficiency prevents memory loss with aging. J. Neurosci. 30: 13816-13821.
[https://doi.org/10.1523/JNEUROSCI.2783-10.2010]

- Yau, J. L. W., McNair, A. J., Noble, J., Seckl, J. R. and Walker, B. R. (2011) The 11β-hydroxysteroid dehydrogenase type 1-specific inhibitor UE1961 improves cognitive function in aged mice. J. Neuroendocrinol. 23: 1252-1259.
- Choudhary, M. I., Yousuf, S., Begum, A., Jahan, S., Sattar, S. A., Shuja, S. and Atta-ur-Rahman. (2011) Structural insights into the inhibition of butyrylcholinesterase by triterpenoids. Bioorg. Med. Chem. 19: 2907-2914.
-
Yogeeswari, P. and Sriram, D. (2005) Betulinic acid and its derivatives: a review on their biological properties. Curr. Med. Chem. 12: 657-666.
[https://doi.org/10.2174/0929867053202214]