Antioxidant Activity and Lifespan-Extending Effects of Sciadopitysin Isolated from Ginkgo biloba Leaves in Caenorhabditis elegans
Abstract
In order to find lifespan extending compound from natural resources, subsequent activity-guided fractionation of Ginkgo biloba leaves (Ginkgoaceae) methanol extract was performed using Caenorhabditis elegans model system. From the most active ethyl acetate soluble fraction, sciadopitysin was isolated, and sciadopitysin showed the antioxidant activities in vitro and in vivo experiments. Sciadopitysin revealed the potent lifespan-promoting activity and it elevated the survival rate of worms against stressful environment including thermal, osmotic and oxidative conditions. As well, sciadopitysin was able to elevate antioxidant enzyme activities of nematodes, and reduced intracellular ROS accumulation of N2 worms in a dose-dependent manner. These results demonstrated that the increased stress tolerance of sciadopitysin-mediated nematode could be attributed to promoted expressions of stress resistance proteins like heat shock protein (HSP-16.2) and superoxide dismutase (SOD-3). And, intestinal lipofuscin level of nematode was decreased by sciadopitysin treatment compared with the control. Moreover it was found that there were no significant changes due to sciadopitysin in aging-related factors including food intake, reproduction, movement and growth. These results suggest that sciadopitysin exerts longevity-promoting activity without affecting above factors. Further study using knockout mutants such as daf-2 and age-1 showed that the sciadopitysin increased the activity of DAF-16 gene which can improve the lifespan of the nematodes.
Keywords:
Ginkgo biloba L., Sciadopitysin, Caenorhabditis elegans, Antioxidant, Lifespan extensionAging is defined by a gradual decline in physiological functions of multiple tissues and organs, during the weakening of organs which have self-renewal and repair abilities.1,2) However, the causes of aging have not yet been comprehensively and quantitatively characterized.3) One of the more currently accepted theories to explain the aging process is free radical theory.4) Excessive free radicals can cause lipid peroxidation, damage biological macromolecules, and lead to cell damage such as nuclear swelling, changes in chromatin structure, decrease or loss of cell replication ability, and slowdown of metabolism; eventually such damage can lead to physical aging and dysfunction, such as inflammation, respiratory disease, and cardiovascular disease. The purpose of this study is to find natural compounds which could be potential candidates for antiaging and antioxidant-related diseases.
Ginkgo biloba leaves (GBL), which are widely used as cosmetics, botanical dietary supplements5) show a variety of biological activities, mostly against cardiovascular and neurological diseases.6,7) GBL is a complex mixture containing different classes of constituents such as flavonol glycosides, terpene trilactones, proanthocyanidines, ginkgolic acids, biflavones, polyflavones and ginkgotoxins.8) Earlier pharmacological studies on G. biloba showed its various bioactivities like neuroprotective, anti-tumor, antioxidant, and amyloid aggregation modulatory effects.9-12)
As a model for study of aging, the natural worm, Caenorhabditis elegans is very attractive due to small size, simple, low-cost, transparency, short life cycle, and variety of transgenic strains.13,14) Because of its simplicity of handling, it is much easier to screen the secondary matabolites of foods and plants which have various biological activities. Furthermore, this worm is not pathogenic, and beyond the ethical boundaries to use in experiments.15-17)
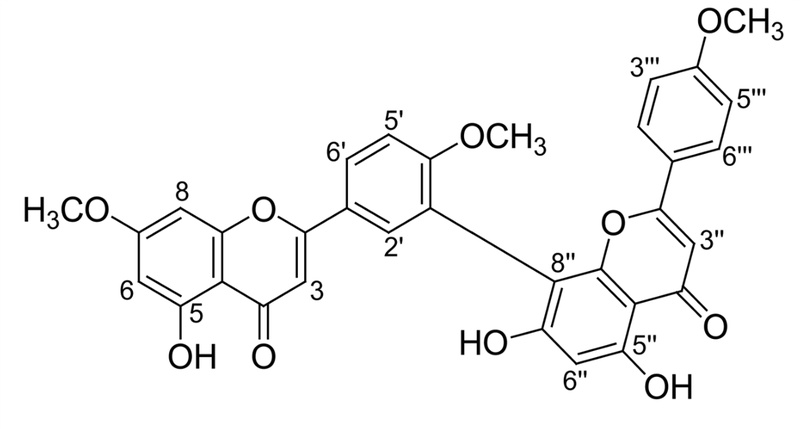
Subsequent activity-guided chromatography of the methanolic extract of G. biloba led to the isolation of sciadopitysin (Fig. 1). In this study, longevity property of sciadopitysin was searched in C. elegans, and several factors that are known to affect aging including antioxidant capability, food intake, development, motility, and lipofuscin levels were examined. Moreover, the genetic requirement for longevity of sciadopitysin was assessed by checking the lifespan of single-gene knockout mutant.
Materials and Methods
General materials – To determine 1H- and 13C-NMR spectra, a JEOL JMN-EX 400 spectrometer was used (Tokyo, Japan). For the column chromatography, Sephadex LH-20 (Fluka, Steinheim, Switzerland) and silica gel (230-400 mesh, Kieselgel 60, Merck, Darmstadt, Germany) were used. Using the precoated silica gel F254 plates, TLC was performed (Merck, Darmstadt, Germany). After TLC, the spots were checked by spaying reagent (10% H2SO4 in ethanol) along with UV. Silica RediSep flash column (Teledyne Isco, Lincoln, US) was used for MPLC. The absorbance to examine the antioxidant effects of samples was determined using microplatereader (Sunrise, Grödig, Austria). The fluorescence of GFP-expressing populations was examined under a fluorescence microscope (Olympus, Tokyo, Japan). Yeast extract and peptone were purchased from Becton, Dickinson and Co. (Sparks, USA). Ascorbic acid, agar, butylated hydroxyanisole (BHA), catalase, 1,1-diphenyl-2-picrylhydrazyl (DPPH), 2’,7’-dichlorodihydrofluoroscein diacetate, and nitroblue tetrazolium (NBT), juglone, xanthine, xanthine oxidase were purchased from Sigma (St. Louis, USA).
Plant Materials – The dried Ginkgo biloba L. leaves were collected from the Woosuk University (Jeonju, Korea), and identified by professor Kim Dae Keun. A voucher specimen of plant was stored in the plant specimen room of College of Pharmacy, Woosuk University (WSU-18-07).
Extraction and Solvent Fractionation – The air dried plant material (3 kg) was extracted three times with methanol at 50℃. The extracts were combined and evaporated in vacuo at 50℃. The resultant methanol extract (450 g) was successively partitioned as n-hexane (130 g) methylene chloride (16 g), ethyl acetate (42 g), n-butanol (129 g) and H2O soluble fractions. These four fractions were tested for the radical scavenging effects on DPPH radical and longevity properties in C. elegans. Among those fractions, the ethyl acetate soluble fraction revealed the most potent activities both DPPH radical scavenging (Fig. 2) and longevity (Fig. 3) properties.
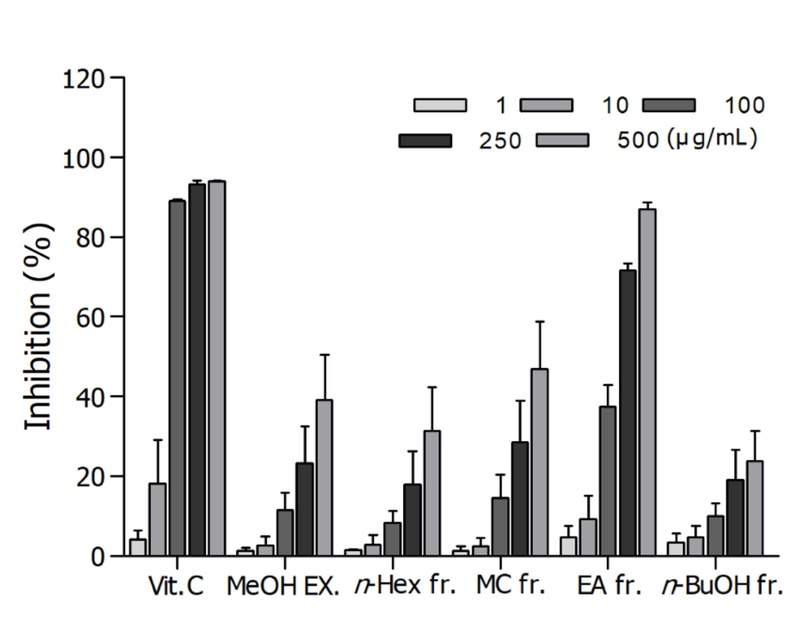
DPPH radical scavenging effects of the MeOH extract and its subsequent fractions from Ginkgo biloba L.
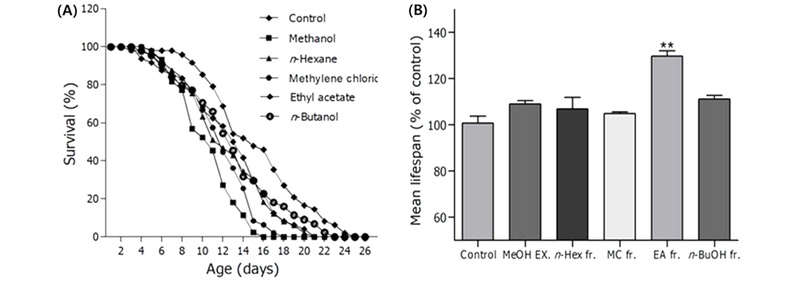
Effects of fractions from G. biloba L. on the lifespan of C. elegans. (A) The mortality of each group was determined by daily counting of surviving and dead worms. (B) The mean lifespan of the N2 worms was calculated from the survival curves in A. Statistical difference between the curves was analyzed by log-rank test. Error bars represent the standard error of mean (S.E.M.). Differences compared to the control were considered significant at **p<0.01 by one-way ANOVA.
Isolation of Sciadopitysin–Sephadex LH-20 column chromatography (MeOH) of the ethyl acetate soluble fraction using methanol as a mobile phase gave eight subfractions (EA1-EA8). Subfraction EA3 (530 mg) was chromatographed by silica gel column (CHCl3 : MeOH, 50:1) and purified on a Sephadex LH-20 (MeOH) to give sciadopitysin (151 mg).
Sciadopitysin-FeCl3 test: positive; 1H-NMR (400 MHz, CD3OD): δ 6.89 (1H, s, H-3), 6.36 (1H, d, J=2.4 Hz, H-6), 6.79 (1H, d, J=2.4 Hz, H-8), 8.08 (1H, d, J=2.4 Hz, H-2'), 7.36 (1H, d, J=8.8 Hz, H-5'), 8.23 (1H, dd, J=2.4, 8.8 Hz, H-6'), 7.00 (1H, s, H-3''), 6.41 (1H, s, H-6''), 7.60 (3H, d, J=9.2 Hz, H-2''', 6'''), 6.93 (3H, d, J=9.2 Hz, H-3''', 5'''), 3.75, 3.78, 3.83 (OCH3, s), 10.97, 12.91, 13.05 (OH, s).
13C-NMR (100 MHz, CD3OD): δ 163.0 (C-2), 103.8 (C-3), 181.9 (C-4), 161.1 (C-5), 98.7 (C-6), 163.6 (C-7), 92.7 (C-8), 157.3 (C-9), 104.7 (C-10), 122.4 (C-1'), 130.9 (C-2'), 121.7 (C-3'), 160.5 (C-4'), 111.7 (C-5'), 128.3 (C-6'), 165.1 (C-2''), 103.2 (C-3''), 182.0 (C-4''), 160.6 (C-5''), 98.1 (C-6''), 162.0 (C-7''), 103.6 (C-8''), 154.3 (C-9''), 103.5 (C-10''), 122.8 (C-1'''), 127.7 (C-2''', 6'''), 114.5 (C-3''', 5'''), 162.2 (C-4'''), 55.5, 55.9, 56.0 (OCH3).
C. elegans Strains and Their Maintenance – The worms were grown at 20°C on nematode growth medium (NGM) agar plates in the presence of Escherichia coli strain OP50, as described previously.18)
Lifespan Assay – C. elegans strains including Bristol N2 (wild-type), CF1553 (SOD-3, muIs84), CL2070 (HSP-16.2, dvls70), DR1572 (daf-2, e1368), TJ1052 (age-1, hx546), GR1307 (age-1, mgDf50), and Escherichia coli OP50 were obtained from the Caenorhabditis Genetic Center (CGC; University of Minnesota, Minneapolis, MN). The nematodes were grown on nematode growth medium (NGM) agar plate with E. coli OP50 (OD600≒0.7) as described previously.19) Sample plates were prepared by adding stock solutions with test samples into autoclaved NGM plates (at 50°C). A final DMSO 0.1% of concentration (v/v) was maintained under all conditions. Age-synchronized nematodes were obtained by embryo isolation. Then, arrested L1 nematodes were transferred to NGM plate in the absence or presence of sciadopitysin (5, 10 μM) and incubated at 20°C. More than 60 animals were used for each independent experiment.
Measurement of Stress Resistance – The age synchronized nematodes were bred on NGM agar plates in the presence or absence of the sample. For the heat tolerance assay, at the adult day, 4 worms were transferred to fresh plates and then incubated at 36°C. The viability was scored over 25 h as previously described.20) Oxidative stress tolerance was assessed as described previously with minor modification.21) In brief, at the 7th adult day, worms were transferred to 96-well plates containing 1 mM juglone, and then survival worms were observed over 35 h. To observe the osmotic effect, the 5 days old of worms were transferred in NGM agar plate containing 500 mM NaCl at 20°C, and determined the survival rates against the osmotic stress following 12 h.22)
Fluorescence Microscopy and Visualization – The transgenic strains CF1553 (SOD-3::GFP reporter) and CL2070 (HSP-16.2::GFP reporter) which were age synchronized were bred in the presence or absence of sciadopitysin. Before the microscopy observation, CL2070 mutants got received thermal shock at 36°C for 2 h and recovered at 20°C for 4 h. On the 3 day of adulthood, both transgenic worms were anesthetized with sodium azide (4%) and mounted on agarose pad. The fluorescence of GFP-expressing populations was examined under a fluorescence microscope. Photographs of the mutants were taken to determine the levels of protein expression, and analyzed using Image J software (Olympus, Tokyo, Japan). All tests were done in triplicate.23)
Assessment of Antioxidant Enzyme Activities–To examine enzymatic activity, the nematodes homogenates were prepared. In brief, the wild-type nematodes were cultured in plate with M9 buffer on third adult day and washed three times. The collected worms were suspended in homogenization buffer (10 mM Tris-HCl, 150 mM NaCl, 0.1 mM EDTA, pH 7.5) and homogenized on ice. The activity of SOD was analyzed by measuring the decolorization of formazan formed by enzymatic reaction between xanthine oxidase and xanthine. The reaction mixture contained 5 μL of worm homogenates, and 120 μL of 0.24 mM nitrobluetetrazolium (NBT) and 0.57 mM xanthine in 10 mM PBS buffer (pH 8.0). After 5 minutes pre-incubation at room temperature, the reaction was begun with adding 100 μL of xanthine oxidase (0.05 U/ml) and incubated for 20 min at 37℃. The reaction was quit by adding 275 μL of 69 mM SDS, and the absorbance was measured at 570 nm. Catalase activity was calculated by spectrophotometry as previously described.24) In brief, the homogenates were blended with 25 mM H2O2, and after 3 min, absorbance was analyzed at 240 nm.
Analysis of Intracellular ROS – Using the molecular probe H2DCF-DA (2',7'-dichlorodihydrofluoroscein diacetate), intracellular ROS in the nematodes was analyzed. Equal number of N2 worms were harvested in the presence or absence of sciadopitysin. On the adult day 4, worms were transferred to 96-well plates containing 50 μM juglone liquid culture for 2 h. After that, 5 worms were exposed to 96-well plates containing 50 μL of M9 buffer. Just after addition of 50 μL of H2DCF-DA solution (25 μM), fluorescence was examined in a microplate fluorescence reader at emission 535 nm and excitation 485 nm.
Lipofuscin Accumulation – C. elegans worms were cultivated from embryo to use the lifespan assay. N2 nematodes were anesthetized with sodium azide to determine the intestinal lipofuscin level, and fluorescence photographs were taken using a fluorescence microscope (Olympus, Tokyo, Japan) on the 8th day of adulthood. Fluorescence intensity was measured by determining pixel intensity in worm’s intestines using Image J software.25)
Measurement of Aging-related Factors – The N2 worms were cultivated on NGM agar plates in the presence or absence of sciadopitysin. On the 4th adult day, worms were transferred to new plates followed by pharynx contractions and movement of worms were calculated under an inverted microscope for 60 seconds. For the growth alteration measurement, photographs were taken of nematodes, and the body length of animals was analyzed by the Olympus software (Olympus, Tokyo, Japan). Reproduction assay was performed as follows. N2 nematodes were raised from embryo for the lifespan assay. Larvae (L4) were individually transferred to new plates every day to separate the parent from the progeny. Counting of the progeny was performed at the larvae 2 or 3 stages. On the 4th and 8th adult days, each nematode was transferred to new plates followed by body movements were observed under the inverted microscope for 20 seconds. Body movements of animals were measured by Olympus image software (Olympus, Tokyo, Japan). All the tests were experimented at least three times.
Data analysis–Using Kaplan-Meier analysis, the lifespan and stress resistance data were plotted, and statistical significance was calculated by log-rank test. Data were showed as mean ± standard error or standard deviation of the mean, as indicated. The statistical significance of differences between treated groups and the control were analyzed by one-way ANOVA.
Results
The total methanol extract was suspended in water and partitioned successively with n-hexane, methylene chloride, ethyl acetate and n-butanol. The ethyl acetate soluble fraction displayed the most significant DPPH free radical scavenging activity among these fractions, and lifespan-extending activity in N2 nematodes (Fig. 2 and 3). Through the consecutive column chromatography using Sephadex LH-20, silica gel, and HPLC led to the isolation of sciadopitysin.
Structure of the Isolated Compound (sciadopitysin) – In the 1H-NMR spectrum, an isolated compound from G. biloba revealed a typical coupling pattern of 1,3,4-trisubstituted aromatic ring at δ 8.23 (1H, dd, J=8.8, 2.4 Hz, H-6'), 7.36 (1H, d, J=8.8 Hz, H-5') and 8.08 (1H, d, J=2.4 Hz, H-2'), and ortho coupling pattern at δ 7.60 (2H, d, J=9.2 Hz, H-2''', 6''') and 6.93 (2H, d, J=9.2 Hz, H-3''', 5'''). Two doublet protons at δ 6.36 (1H, d, J=2.4 Hz, H-6) and 6.79 (1H, d, J=2.4 Hz, H-8), and three singlet protons at δ 7.00 (3H, s, H-3''), 6.89 (3H, s, H-3) and 6.41 (3H, s, H-6'') were observed in the aromatic region. In addition, three methoxyl groups at δ 3.75, 3.78, 3.83, and three hydroxy protons at δ 10.97, 12.91, 13.05 were detected in the 1H-NMR spectrum. The 13C-NMR spectrum of the compound showed 31 carbon signals including two carbonyl carbons at δ 182.0 (C-4''), 181.9 (C-4), 10 oxygen-bearing olefinic carbons (δ 165.1, 163.6, 163.0, 162.8, 162.0, 161.1, 160.5, 157.3 and 154.4), and three methoxyl carbons. From above evidences, the structure of the compound was deduced as one of the biflavonoids previously isolated from the G. biloba. By direct comparison of its spectral data with published data, the compound was characterized to be sciadopitysin.26)
Antioxidative Effects
Fractions of G. biloba L. - DPPH radical scavenging effect - DPPH radical scavenging effect of vitamin C, the positive control showed the IC50 value of 27.27 μg/ml. Among four solvent fractions, ethyl acetate fraction of G. biloba L. exhibited the most potent scavenging effect does-dependently on DPPH radical with the IC50 value of 134.9 μg/ml (Fig. 2).
Anti-aging Activities
Effects of Fractions on the Lifespan in N2 Worms - Lifespan assays were examined with wild-type N2 worms to determine the longevity properties of G. biloba L. Ethyl acetate soluble fraction revealed the most significant lifespan extending effect among four fractions. In addition, the ethyl acetate fraction mediated worms showed the significant increase of the estimated mean life compared to the control worms (27.36%, p<0.01) (Fig. 3B). The mean life duration was 12.14±0.7 days for control worms and 15.45±0.7 for the worms fed the ethyl acetate fraction (Fig. 3, Table I).
Effects of sciadopitysin on the lifespan of C. elegans – In order to determine the lifespan extension property of sciadopitysin, lifespan assay was performed with wild-type N2 worms. sciadopitysin showed the longevity property in a dose dependent manner (Fig. 4A). sciadopitysin-mediated worms showed the significant increase of the estimated mean life compared to the control worms (33.53% at 10 µM, p<0.001) (Fig. 4B, Table II). The mean life durations for control and sciadopitysin-mediated worms were 17.04±0.8 and 18.96±1.1 (10 µM) days, respectively (Fig. 4, Table II).
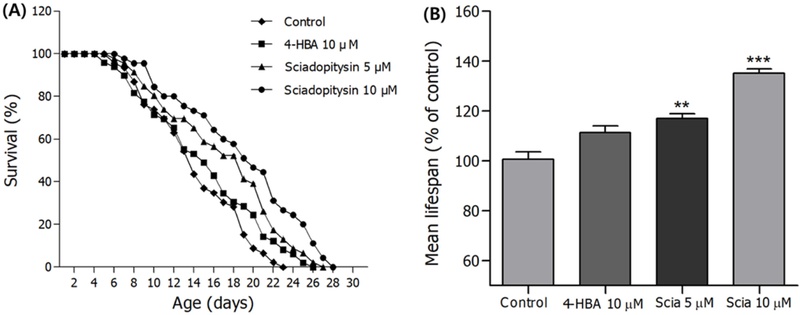
Effects of sciadopitysin from G. biloba L. on the lifespan of C. elegans. (A) The mortality of each group was determined by daily counting of surviving and dead animals. (B) The mean lifespan of the N2 worms was calculated from the survival curves in (A). Statistical difference between the curves was analyzed by log-rank test. Error bars represent the standard error of mean (S.E.M.). Differences compared to the control were considered significant at **p<0.01 and ***p<0.001 by one-way ANOVA.
Effects of sciadopitysin on the stress tolerance of C. elegans – The effects of sciadopitysin on three different kinds of stress conditions such as osmotic, thermal and oxidative stresses using wild-type C. elegans were determined. In the case of hypertonic stress assay, sciadopitysin-mediated N2 worms exhibited enhanced resistance to osmotic stress (Fig. 5A). sciadopitysin exposure induced potent increases in thermotolerance, and consequently raised survival rate of worms (Fig. 5B, Table III). Besides, sciadopitysin exposure increased the maximum lifespan of nematodes by 40.63% (10 µM, p<0.001). Furthermore, it was revealed that sciadopitysin-mediated nematodes survived longer than control worms by 36.13% under oxidative stress conditions induced by 1 mM juglone in 10 μM (p<0.001, Fig. 5C, Table III).
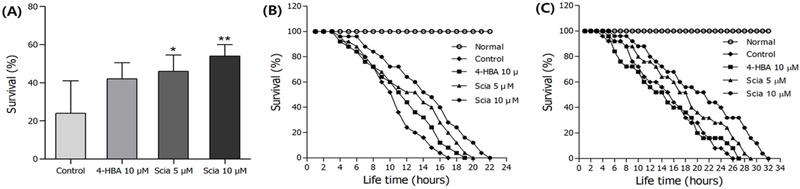
Effects of sciadopitysin on the stress tolerance of wild-type N2 nematodes. (A) Tolerance of osmotic stress survival rates were determined following 12 h. (B) To assess heat tolerance, worms were incubated at 36℃ and then their viability was checked. (C) For the oxidative stress assays, nematodes were transferred to well plate containing juglone-treated culture, and then their were scored for viability. Statistical difference between the curves was analyzed by log-rank test. Error bars represent the standard error of mean (S.E.M.). Differences compared to the control were considered significant at *p<0.05, **p<0.01 and ***p<0.001 by one-way ANOVA.
Effects of Sciadopitysin on the Stress Resistance Proteins of C. elegans – To investigate whether sciadopitysin-mediated increased stress resistances were due to the regulation of gene which are related to stress-response, SOD-3 and HSP-16.2 expressions were quantified using transgenic strains such as CF1553 and CL2070, respectively. It was revealed that sciadopitysin-mediated CF1553 nematodes exhibited significantly higher SOD-3::GFP intensity (52.69% at 10 μM, p<0.001), compared to the untreated control worms (Fig. 6). The CL2070 worms bearing HSP-16.2::GFP reporter gene got stressed by thermal shock at 36℃ for 2 h, and after recovering at 20℃ for 4 h, fluorescence intensities were quantified. This heat shock-derived HSP-16.2::GFP expression more increased by 10 μM of sciadopitysin-mediated about 24.53% (p<0.001, Fig. 7).
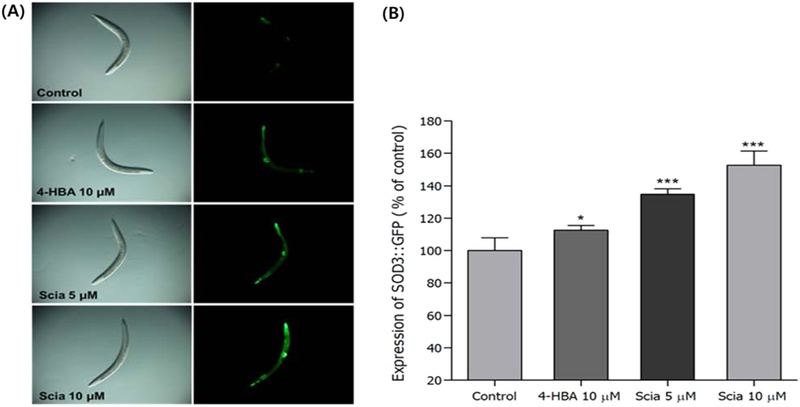
Effects of sciadopitysin on the expression of SOD-3(CF1553) was determined using transgenic nematodes. Mean GFP-expressing intensity of CF1553 mutants were expressed as mean S.E.M. of values from 70-80 worms per each experiment. (A) Expressions of CF1553 nematodes in the presence or absence of sciadopitysin (B) The GFP-expressing intensity was quantified using software by determining mean pixel intensity. Images of SOD-3::GFP Data are expressed as the mean±standard deviation of three independent experiments. Differences compared to the control were considered significant at *p<0.05 and ***p<0.001 by one-way ANOVA.
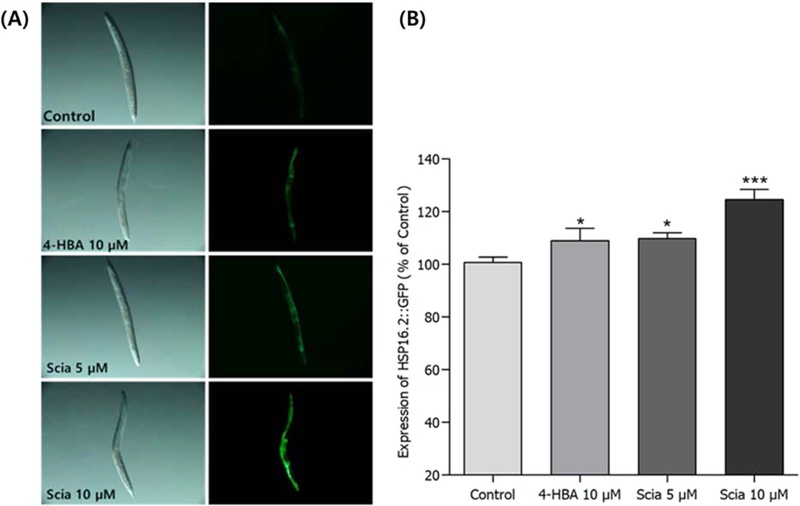
Effects of sciadopitysin on the expression of HSP-16.2(CL2070) was determined using transgenic nematodes. Mean GFP-expressing intensity of CL2070 mutants were expressed as mean ± S.E.M. of values from 70-80 worms per each analysis. (A) Expressions of CL2070 nematodes in the presence or absence of sciadopitysin (B) The GFP-expressing intensity was quantified using software by determining mean pixel intensity. Image of HSP-16.2::GFP Data are expressed as the mean ± standard deviation of three independent experiments. Differences compared to the control were considered significant at **p<0.01, ***p<0.001 by one-way ANOVA.
Effects of Sciadopitysin on the Antioxidant Enzyme Activities in C. elegans – In order to verify the mechanism of sciadopitysin on increasing lifespan and stress resistance of N2 worms, effects of sciadopitysin on the antioxidant enzyme activity was investigated. The SOD enzymatic activity was spectrophotometrically experimented using prepared worm homogenate. It was observed that sciadopitysin-mediated elevated SOD activity of nematodes significantly by 28.94% at 10 μM (p<0.001, Fig. 8A). From the measuring of the residual H2O2 concentration as determined by a spectrophotometric method, catalase activity was calculated. sciadopitysin significantly elevated catalase activity of N2 worms by 60.74% (10 μM, p<0.001, Fig. 8B).
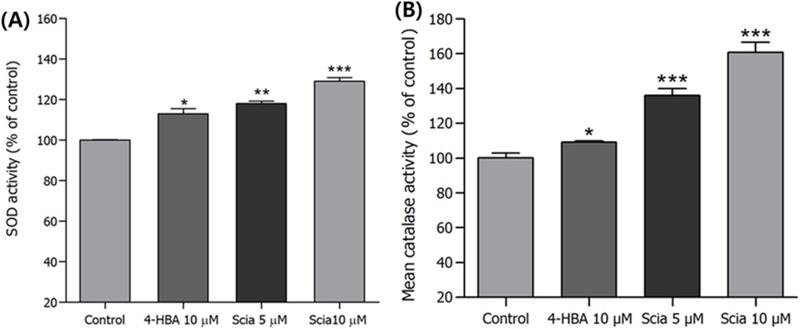
Effects of sciadopitysin on the antioxidant enzyme activity of wild type N2 nematodes. (A) SOD activity was showed as a percentage of superoxide-scavenged amount per control. (B) Average catalase activity of each group was determined by seconds counting. Differences compared to the control were considered significant at *p<0.05, **p<0.01 and ***p<0.001 by one-way ANOVA.
Effects of Sciadopitysin on the Intracellular ROS – Intracellular ROS level of sciadopitysin-mediated worms were quantified compared to untreated controls. Fig. 9 shows that sciadopitysin-mediated worms effectively diminished the production of ROS by 23.59% (10 μM, p<0.001), respectively, compared to solvent-treated control worms. Since sciadopitysin has strong antioxidant capacity, it could be assumed that radical scavenging effects and increase of antioxidant enzyme activities of sciadopitysin result in the attenuation of intracellular ROS level.
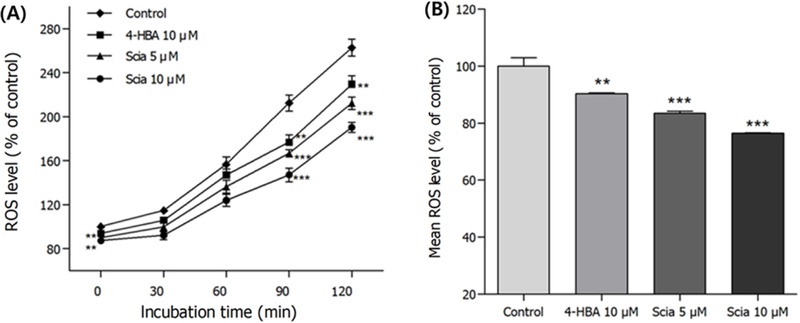
Effects of sciadopitysin on the intracellular ROS levels of wild-type nematodes. Intracellular ROS accumulation was examined in a microplate fluorescence reader at emission 535 nm and excitation 485 nm. (A) Plates were read 0 min for 120 min. (B) The average percentage of intracellular ROS accumulation were indicated. Differences compared to the control were considered significant at **p<0.01 and ***p<0.001 by one-way ANOVA.
Effects of Sciadopitysin on the Lipofuscin Accumulation–As one of endogenous markers of cellular damage, lipofuscin is known one during aging in many organisms, like wild-type N2 nematode. Lipofuscin is known as granular yellow-brown pigment granules which are composed of lipid-containing remnant of lysosomal digestion. The autofluorescence level of lipofuscin can be measured by fluorescence microscope. sciadopitysin-mediated worms showed significant decrease of fluorescence intensity from intestinal lipofuscin by 15.36% at 10 μM, respectively, compared with the control (p<0.001, Fig. 10).
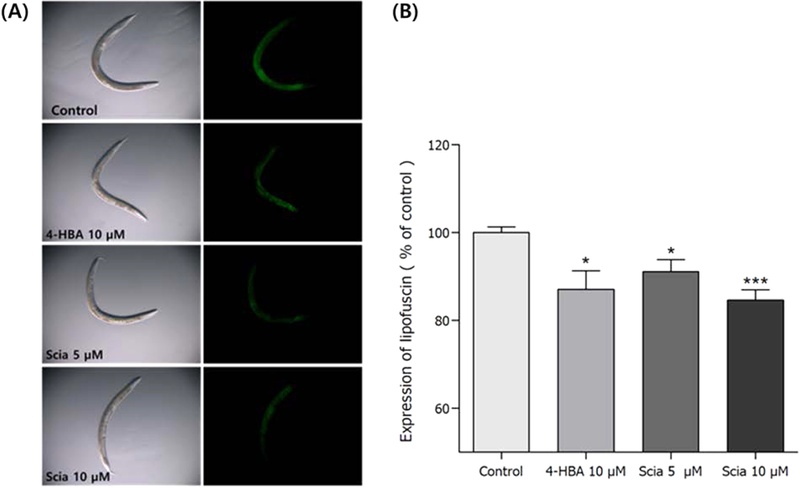
Effects of sciadopitysin on the lipofuscin accumulation in wild-type N2 nematodes. (A) Image of intestinal autofluorescence from lipofuscin accumulation of worms on the 8th adult days. (B) Fluorescence intensity was measured by determining pixel intensity in worm’s intestines using Image J software. Mean fluorescence intensity of lipofuscin accumulation was expressed as mean ± S.E.M. Differences compared to the control were considered significantat *p<0.05 and ***p<0.001 by one-way ANOVA.
Effects of Sciadopitysin on the Aging-related Factors of C. elegans – To verify the mechanism of sciadopitysin on the lifespan of nematodes, sciadopitysin-induced changes in parameters of aging-related factors like progeny, food intake, locomotion and body length were observed. It was not detected any differences between sciadopitysin-mediated and control nematodes on the reproduction rate, pumping rate, movement and body length (Fig. 11).
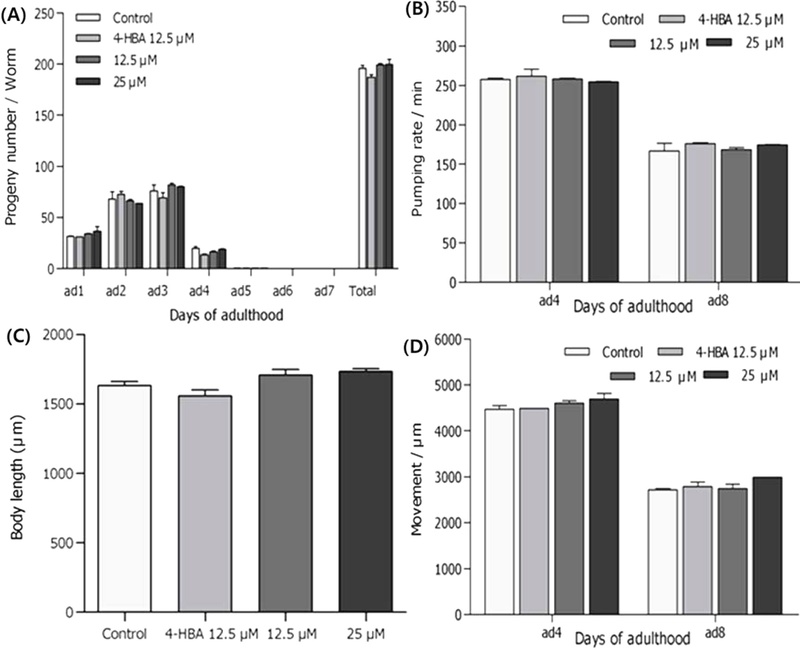
Effects of sciadopitysin on the aging-related factors of C. elegans. (A) Adult 1 to 7 and total reproductive progeny were counted. (B) On the 4th adult days, the pharynx contraction rates were measured. (C) The body movements were observed under the inverted microscope for 20 seconds. (D) For the growth alteration measurement, photographs were taken of nematodes, and the body length of animals was analyzed. Data are expressed as the mean ± S.E.M. of three independent experiments.
Target Genes of Sciadopitysin-mediated Longevity Properties – In this study, it was further assayed the involvement of insulin/IGF-1 signaling pathway in the sciadopitysin-mediated longevity properties to see possible underlying mechanism. Another lifespan assays were carried out again using several null mutant strains, including DR1572 (daf-2, e1368), TJ1052 (age-1, hx546), GR1307 (daf-16 mgDf50). Sciadopitysin significantly increased the lifespan of daf-2 and age-1 mutants, but daf-16 mutant was not affected by the sciadopitysin (Fig. 12, Table III). These results imply that these genes are required for the sciadopitysin-mediated longevity.
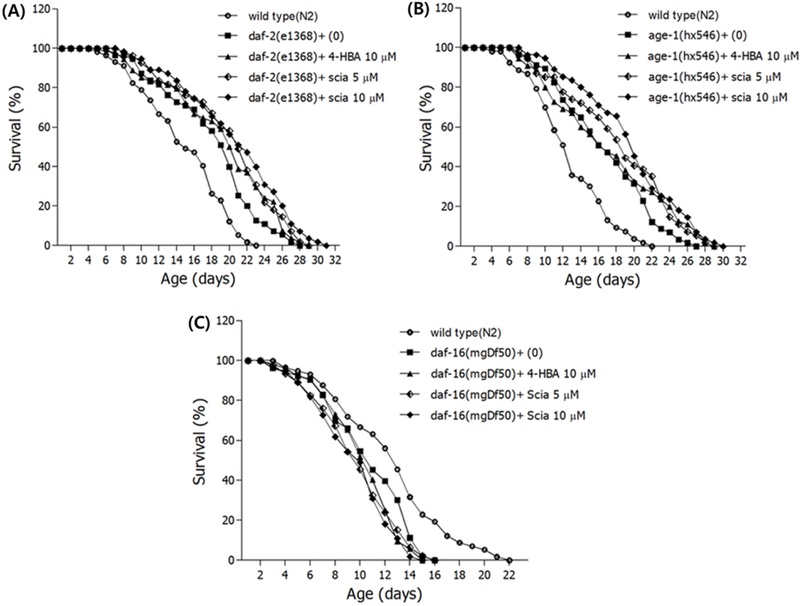
Target genes of sciadopitysin-mediated longevity properties. (A) The survival curves of wild-type and daf-2 (e1368) mutant nematodes treated with or without 5 and 10 μM of sciadopitysin (B) The survival curves of wild-type and age-1 (hx546) mutant nematodes treated with or without 5 and 10 μM of sciadopitysin (C) The survival curves of wild-type and daf-16 (mgDf50) mutant nematodes treated with or without 5 and 10 μM of sciadopitysin. All experiments were done in triplicate independently.
Discussion
During the ongoing investigation to find lifespan extension compounds from natural materials, sciadopitysin was isolated from the methanol extract of G. biloba as an active constituent using the C. elegans model system. This system offered as a well-established model for aging research including various lifespan-extending mechanism study.27)
Sciadopitysin, isolated from G. biloba, was reported lots of its biological effects including UDP-glucuronosyltransferases, cGMP-phosphodiesterase-5, and phosphatase of regenerating liver-3 inhibitory effects,28-30) and protective effects of myocardial necrosis and methylglyoxal-induced cytotoxicity,31,32) along with anti-Alzheimer's disease.33) Although sciadopitysin has shown various effects, but its lifespan extension activity on C. elegans is unknown.
The nematode which is wild-type strain N2 worms treated with sciadopitysin showed longevity property than non-treated ones under a normal condition. Recently, much research have been reported on the close relationship between lifespan and stress resistance.34-36) Applications of the three types of stress significantly decreased the average lifespan of the worms, but the treatment of sciadopitysin prolonged the reduced lifespan in a dose-dependent manner. This result suggested that sciadopitysin is capable of increasing survival rates of C. elegans partly by improving stress resistance. As a part of the effort to elucidate the lifespan-extending mechanism of sciadopitysin, the scavenging activity of antioxidant enzymes such as SOD and catalase that affect free radical and ROS levels were calculated in relation to the increased oxidative stress resistance. In the wild-type strain N2 worms, sciadopitysin intake increased the activity levels of the two endogenous antioxidant enzymes SOD and catalase dose-dependently. These results are in agreement with the earlier report that sciadopitysin protects osteoblast function via its antioxidant activity in MC3T3-E1 cells.37) In addition, the intracellular ROS levels of the N2 worms were measured, showing dose-dependent reduction in ROS accumulation by the sciadopitysin treatment. These results supported a hypothesis that the antioxidant effect of sciadopitysin contributes to the lifespan elongation of C. elegans in part via the up-regulation of endogenous antioxidant enzymes, reinforcing the close relationship between aging and free radical/ROS levels.38) In addition, expression levels of the stress resistance proteins SOD-3 and HSP-16.2 were determined in transgenic strains using green fluorescent protein (GFP)-based reporters. Higher fluorescence intensity was observed when the two types of transgenic worms were grown in the presence of sciadopitysin, indicating that the compound induced SOD-3 and HSP-16.2 expressions.39) This result confirmed that the anti-aging mechanism of sciadopitysin involves an endurance in stress resistances through enzyme induction. Lipofuscin, autofluorescent granules in organisms, accumulates during normal aging. So the autofluorescent intensity of which enables estimation of the aging process.40)
Treatment of sciadopitysin decreased the level of intestinal lipofuscin of worms compared to that of non-treated controls presuming, supporting its longevity activity. Other aging-related factors are known to affect the aging process such as reproduction, food intake, growth, and movement.41,42) However, adding sciadopitysin made no significant differences to their offspring, the pumping of pharynx, body movements, or body length. These results indicated the independence of such factors from the lifespan-extending effects observed for treatment of sciadopitysin.
Several genes, like DAF-2 and AGE-1, are related to the activation of DAF-16 gene which can increase the lifespan. Loss of function mutations of daf-2 and age-1 extend lifespan in a daf-16-dependent manner.43-45) Sciadopitysin-treatment significantly increased the lifespan of daf-2 and age-1 mutants, but daf-16 mutant was not affected by the compound, supporting sciadopitysin might increase the activity of DAF-16 gene.
Conclusions
In order to search for lifespan extending compounds from natural resources, subsequent activity-oriented chromatography of G. biloba methanolic extract was performed using C. elegans model system. Repetitive chromatography using silica gel and Sephadex LH-20, and purifying with HPLC led to the isolation of a compound, sciadopitysin from the most active ethyl acetate soluble fraction.
Sciadopitysin-treatment extended the lifespan of N2 worms than the controls. Under osmotic, thermal and juglone-induced oxidative stresses sciadopitysin-fed worms also survived much longer than the control group. Sciadopitysin was able to increase SOD and catalase activities of worms in a dose dependent manner. Furthermore, the intracellular ROS accumulations also decreased by sciadopitysin exposure. Effort to elucidate the mechanism of such action suggested that stress tolerances of worms increased upon treatment with sciadopitysin. Expressions of the proteins SOD-3 and HSP-16.2 were induced by the sciadopitysin treatment in the transgenic strains, supporting the proposed mechanism. Another set of supportive result was observed when accumulation level of lipofuscin, autofluorescent marker of aging-were significantly decreased in the sciadopitysin-fed worms. Further study using knockout mutants showed that the sciadopitysin increased the activity of DAF-16 gene which can improve the lifespan of the nematodes. Consequently, sciadopitysin could be a useful and attractive candidate for an anti-aging related resource.
References
-
Li, G., Gong, J., Liu, J., Liu, J., Li, H., Hsu, A. L., Liu, J. and Xu, X. S. (2019) Genetic and pharmacological interventions in the aging motor nervous system slow motor aging and extend lifespan in C. elegans. Sci. Adv. 5: e5041.
[https://doi.org/10.1126/sciadv.aau5041]

-
Xu, H. J., Hao, X. L., Qiao, D. L., Xia, L. B., Chen, R., He, X. M., Gu, F. L. and Wei, C. B. (2019) Effects of Dendrobium huoshanense polysaccharides on antioxidant capacity, mucosal barrier integrity and inflammatory responses in an aging rat ileal model. Biotechnol. Biotechnol. Equip. 33: 1444-1452.
[https://doi.org/10.1080/13102818.2019.1674187]

-
Li, S. T., Zhao, H. Q., Zhang, P., Liang, C. Y., Zhang, Y. P., Hsu, A. L. and Dong, M. Q. (2019) DAF-16 stabilizes the aging transcriptome and is activated in mid‐aged Caenorhabditis elegans to cope with internal stress. Aging Cell 18: e12896.
[https://doi.org/10.1111/acel.12896]

-
Kasote, D. M., Jayaprakasha, G. K. and Patil, B. S. (2019) Leaf disc assays for rapid measurement of antioxidant activity. Sci. Rep. 9: 1884.
[https://doi.org/10.1038/s41598-018-38036-x]

-
Huang, X. F., Zhang, S. Z., You, Y. Y., Zhang, N., Lu, H., Daugherty, A. and Xie, X. J. (2019) Ginkgo biloba extracts prevent aortic rupture in angiotensin II-infused hypercholesterolemic mice. Acta Pharmacol. Sin. 40: 192-198.
[https://doi.org/10.1038/s41401-018-0017-7]

-
Piazza, S., Pacchetti, B., Fumagalli, M., Bonacina, F., Dell’Agli, M. and Sangiovanni, E. (2019) Comparison of two Ginkgo biloba L. extracts on oxidative stress and inflammation markers in human endothelial cells. Mediators Inflamm. 2019: e6173893.
[https://doi.org/10.1155/2019/6173893]

-
Wu, Y., Zhou, Q., Huang, S., Wang, G. and Xu, L. (2019) SNP development and diversity analysis for Ginkgo biloba based on transcriptome sequencing. Trees 33: 587-597.
[https://doi.org/10.1007/s00468-018-1803-z]

-
Waidyanatha, S., Mutlua, E., Gibbsb, S., Stiffler, B., Andre, J., Burback, B. and Rider, C. V. (2019) Systemic exposure to Ginkgo biloba extract in male F344/NCrl rats: relevance to humans. Food Chem. Toxicol. 131: e110589.
[https://doi.org/10.1016/j.fct.2019.110586]

-
Al-Adwani, D. G., Renno, W. M. and Orabi, K. Y. (2019) Neurotherapeutic effects of Ginkgo biloba extract and its terpene trilactone, ginkgolide B, on sciatic crush injury model: A new evidence. PLoS One 14: e0226626.
[https://doi.org/10.1371/journal.pone.0226626]

-
Verma, S., Sharma, S., Ranawat, P. and Nehru, B. (2020) Modulatory effects of Ginkgo biloba against amyloid aggregation through induction of heat shock proteins in aluminium-induced neurotoxicity. Neurochem. Res. 45: 1-26.
[https://doi.org/10.1007/s11064-019-02940-z]

- Cao, C. J., Su, Y., Sun, J., Wang, G. Y., Jia, X. Q., Chen, H. S. and Xu, A. H. (2019) Anti-tumor effect of Ginkgo biloba exocarp extracts on B16 melanoma bearing mice involving PI3K/Akt/HIF-1α/VEGF signaling pathways. Iran J. Pharm. Res. 18: 803-811.
-
Singh, S. K., Srivastav, S., Castellani, R. J., Plascencia-Villa, G. and Perry, G. (2019) Neuroprotective and antioxidant effect of Ginkgo biloba extract against AD and other neurological disorders. Neurotherapeutics 16: 666-674.
[https://doi.org/10.1007/s13311-019-00767-8]

-
Wang, H., Webster, P., Chen, L. and Fisher, A. L. (2019) Cell-autonomous and non-autonomous roles of daf-16 in muscle function and mitochondrial capacity in aging C. elegans. Aging 11: 2295-2311.
[https://doi.org/10.18632/aging.101914]

-
Liu, J., Zhang, B., Lei, H., Feng, Z., Liu, J., Hsu, A. H. and Xu, S. X. Z. (2013) Functional aging in the nervous system contributes to age-dependent motor activity decline in C. elegans. Cell Metab. 18: 392-402.
[https://doi.org/10.1016/j.cmet.2013.08.007]

-
Sugawara, T., Furuhashi, T., Shibata, K., Abe, M., Kikuchi, K., Arai, M. and Sakamoto, K. (2019) Fermented product of rice with Lactobacillus kefiranofaciens induces anti-aging effects and heat stress tolerance in nematodes via DAF-16. Biosci. Biotechnol. Biochem. 83: 1484-1489.
[https://doi.org/10.1080/09168451.2019.1606696]

-
Ayuda-Durán, B., González-Manzano, S., Miranda-Vizuete, A., Sánchez-Hernández, E., Romero, M. R., Dueñas, M., Santos-Buelga, C. and González-Paramás, A. M. (2019) Exploring target genes involved in the effect of quercetin on the response to oxidative stress in Caenorhabditis elegans. Antioxidants 8: 585.
[https://doi.org/10.3390/antiox8120585]

-
Lithgow, G. J., White, T. M., Melov, S. and Johnson, T. E. (1995) Thermotolerance and extended lifespan conferred by single-gene mutations and induced by thermal stress. Proc. Natl. Acad. Sci. USA. 92: 7540-7544.
[https://doi.org/10.1073/pnas.92.16.7540]

-
Leiser, S. F., Miller, H., Rossner, R., Fletcher, M., Leonard, A., Primitivo, M., Rintala, N., Ramos, F. J., Miller, D. L. and Kaeberlein, M. (2015) Cell nonautonomous activation of flavin-containing monooxygenase promotes longevity and health span. Science 350: 1375-1378.
[https://doi.org/10.1126/science.aac9257]

-
Zhang, H. L., Jia, F., Li, M., Yu, F., Zhou, B., Hao, Q. H. and Wang, X. L. (2019) Endophytic Bacillus strains isolated from alfalfa (Medicago sativa L.) seeds: enhancing the lifespan of Caenorhabditis elegans. Lett. Appl. Microbiol. 68: 226-233.
[https://doi.org/10.1111/lam.13102]

-
Lee, E. Y., Shim, Y. H., Chitwood, D. J., Hwang, S. B., Lee, J. and Paik, Y. K. (2005) Cholesterol-producing transgenic Caenorhabditis elegans lives longer due to newly acquired enhanced stress resistance. Biochem. Biophys. Res. Commun. 328: 929-936.
[https://doi.org/10.1016/j.bbrc.2005.01.050]

-
Mekheimer, R. A., Sayed, A. A. A. and Ahmed, E. A. (2012) Novel 1,2,4-triazolo[1,5-a] pyridines and their fused ring systems attenuate oxidative stress and prolong lifespan of Caenorhabditis elegans. J. Med. Chem. 55: 4169-4177.
[https://doi.org/10.1021/jm2014315]

-
Pujol, N., Zugasti, O., Wong, D., Couillault, C., Kurz, C. L., Schulenburg, H. and Ewbank, J. J. (2008) Anti-fungal innate immunity in C. elegans is enhanced by evolutionary diversification of antimicrobial peptides. PLoS Pathog. 4: e1000105.
[https://doi.org/10.1371/journal.ppat.1000105]

-
Waters, J. C. (2009) Accuracy and precision in quantitative fluorescence microscopy. J. Cell Biol. 185: 1135-1148.
[https://doi.org/10.1083/jcb.200903097]

-
Aebi, H. (1984) Catalase in vitro. Methods Enzymol. 105: 121-126.
[https://doi.org/10.1016/S0076-6879(84)05016-3]

-
Brunk, U. T. and Terman, A. (2002) Lipofuscin: mechanisms of age-related accumulation and influence on cell function. Free Radic. Biol. Med. 33: 611-619.
[https://doi.org/10.1016/S0891-5849(02)00959-0]

- Kang, S. S., Kim, J. S., Son, K. H., Kim, H. P. and Chang, H. W. (1990) Siadopitysin, a biflavonoid from Ginkgo biloba, inhibits phospholipase A2. Planta Med. 56: 540-542.
-
Yoshida, M., Satoh, A., Lin, J. B., Mills, K. F., Sasaki, Y., Rensing, N., Wong, M., Apte, R. S. and Imai, S. I. (2019) Extracellular vesicle-contained eNAMPT delays aging and extends lifespan in mice. Cell Metab. 30: 329-342.
[https://doi.org/10.1016/j.cmet.2019.05.015]

- Wang, X. X., Hou, J., Ning, J., Pan, Y. Q., Hong, M. and Guo, B. (2016) Inhibition of sciadopitysin against UDP-glucuronosyltransferases. Yao Xue Xue Bao 51: 749-755.
-
Dell'Agli, M., Galli, G. V. and Bosisio, E. (2006) Inhibition of cGMP-phosphodiesterase-5 by biflavones of Ginkgo biloba. Planta Med. 72: 468-470.
[https://doi.org/10.1055/s-2005-916236]

-
Choi, S. K., Oh, H. M., Lee, S. K., Jeong, D. G., Ryu, S. E., Son, K. H., Han, D. C., Sung, N. D., Baek, N. I. and Kwon, B. M. (2006) Biflavonoids inhibited phosphatase of regenerating liver-3 (PRL-3). Nat. Prod. Res. 20: 341-346.
[https://doi.org/10.1080/14786410500463312]

- Cai, Y. and Li, Y. (2019) Protective effect of sciadopitysin against isoproternol-induced myocardial infarction in rats. Pharmacology 27: 1-9.
-
Suh, K. S., Chon, S. and Choi, E. M. (2018) The protective effects of sciadopitysin against methylglyoxal-induced cytotoxicity in cultured pancreatic β-cells. J. Appl. Toxicol. 38: 1104-1111.
[https://doi.org/10.1002/jat.3620]

-
Gu, Q., Li, Y., Chen, Y., Yao, P. and Ou, T. (2013) Sciadopitysin: active component from Taxus chinensis for anti-Alzheimer's disease. Nat. Prod. Res. 27: 2157-2160.
[https://doi.org/10.1080/14786419.2013.790031]

-
Wang, Z., Komatsu, T., Ohata, Y., Watanabe, Y., Yuan, Y., Yoshii, Y., Park, S., Mori, R., Satou, M., Kondo, Y., Shimokawa, I. and Chiba, T. (2019) Effects of rikkunshito supplementation on resistance to oxidative stress and lifespan in mice. Geriatr. Gerontol. Int.
[https://doi.org/10.1111/ggi.13848]

-
Prasanth, M. I., Brimson, J. M., Chuchawankul, S., Sukprasansap, M. and Tencomnao, T. (2019) Antiaging, stress resistance, and neuroprotective efficacies of Cleistocalyx nervosum var. paniala fruit extracts using Caenorhabditis elegans model. Oxid. Med. Cell Longev.
[https://doi.org/10.1155/2019/7024785]

-
Dilberger, B., Baumanns, S., Schmitt, F., Schmiedl, T., Hardt, M., Wenzel, U. and Eckert, G. P. (2019) Mitochondrial oxidative stress impairs energy metabolism and reduces stress resistance and longevity of C. elegans. Oxid. Med. Cell Longev.
[https://doi.org/10.1155/2019/6840540]

-
Suh, K. S., Lee, Y. S., Kim, Y. S. and Choi, E. M. (2013) Sciadopitysin protects osteoblast function via its antioxidant activity in MC3T3-E1 cells. Food Chem. Toxicol. 58: 220-227.
[https://doi.org/10.1016/j.fct.2013.04.028]

-
Davalli, P., Mitic, T., Caporali, A., Lauriola, A. and D'Arca, D. (2016) ROS, cell senescence, and novel molecular mechanisms in aging and age-related diseases. Oxid. Med. Cell Longev.
[https://doi.org/10.1155/2016/3565127]

-
Azevedo, B. C., Roxo, M., Borges, M. C., Peixoto, H., Crevelin, E. J., Bertoni, B. W., Contini, S. H. T., Lopes, A. A., Franca, S. C., Pereira, A. M. and Wink, M. (2019) Antioxidant activity of an aqueous leaf extract from Uncaria tomentosa and its major alkaloids mitraphylline and isomitraphylline in Caenorhabditis elegans. Molecules 24: 3299.
[https://doi.org/10.3390/molecules24183299]

-
Singh Kushwaha, S., Patro, N. and Patro, I. K. (2019) A sequential study of age-related lipofuscin accumulation in hippocampus and striate cortex of rats. Ann. Neurosci. 25: 223-233.
[https://doi.org/10.1159/000490908]

-
Zhou, S. J., Zhao, M. J., Yang, Y. H., Guan, D., Li, Z. G., Ji, Y. D., Zhang, B. L., Shang, X. J., Xiong, C. L. and Gu, Y. Q. (2020) Age-related changes in serum reproductive hormone levels and prevalence of androgen deficiency in Chinese community-dwelling middle-aged and aging men: two cross-sectional studies in the same population. Medicine (Baltimore) 99: e18605.
[https://doi.org/10.1097/MD.0000000000018605]

-
Gopinath, B., Liew, G., Kifley, A., Lewis, J. R., Bondonno, C., Joachim, N., Hodgson, J. M. and Mitchell, P. (2018) Association of dietary nitrate intake with the 15-year incidence of age-related macular degeneration. J. Acad. Nutr. Diet. 118: 2311-2314.
[https://doi.org/10.1016/j.jand.2018.07.012]

-
Zhang, Z., Zhou, Y., Fan, H., Billy, K. J., Zhao, Y., Zhan, X., Yang, L. and Jia, Y. (2019) Effect of lycium barbarum polysaccharides on health and aging of C. elegans depends on daf-2/daf-16. Oxid. Med. Cell Longev.
[https://doi.org/10.1155/2019/6379493]

-
Saier, C., Büchter, C., Koch, K. and Wätjen, W. (2018) Polygonum multiflorum extract exerts antioxidative effects and increases lifespan and stress resistance in the model organism Caenorhabditis elegans via DAF-16 and SIR-2.1. Plants 7: 60.
[https://doi.org/10.3390/plants7030060]

-
Yuan, Y., Kang, N., Li, Q., Zhang, Y., Liu, Y. and Tan, P. (2019) Study of the effect of neutral polysaccharides from rehmannia glutinosa on lifespan of Caenorhabditis elegans. Molecules 24: 4592.
[https://doi.org/10.3390/molecules24244592]