
Lifespan-extending Effects of Ikarisoside A from Epimedium koreanum in Caenorhabditis elegans
Abstract
This experiment was conducted to find anti-aging compounds using nematodes from natural products. After separating the ikarisoside A from Epimedium koreanum Nakai (Berberidaceae), an anti-aging experiment was followed using a Caenorhabditis elegans model. The nematodes treated with ikarisoside A showed lifespan extension effect under general culture conditions. In addition, ikarisoside A increased the survival rate of C. elegans in stressful environments such as heat and osmotic conditions. To study the expression level of HSP-16.2, we used a transgenic strain including CL2070, and ikarisoside A-treated worms had a higher HSP-16.2::GFP intensity compared to untreated CL2070 worms. Nematodes fed the ikarisoside A showed reduced lipofuscin accumulation compared to control worms. Furthermore, there were no significant changes due to ikarisoside A in aging-related factors such as food intake, exercise, reproduction, and growth, indicating that ikarisoside A induces longevity activities without affecting these factors. These results indicate that ikarisoside A can affect longevity of C. elegans under both normal and stress conditions through the expression of stress-resistant proteins.
Keywords:
Epimedium koreanum Nakai, Ikarisoside A, Caenorhabditis elegans, Lifespan-extending effect, Stress resistantAging is an inevitable phenomenon caused by a number of factors within a living organism. Biologically, the survival of an organism is an ongoing struggle between biochemical damage and recovery. Many molecules, cells, biochemical pathways, and networks determine survival and lifespan. Depending on the frequency, intensity, duration, energy consumption costs, and other metabolic disorders of stress occurring in vivo, the consequences of stress can be beneficial or harmful, but excessive stress can promote aging.1,2,7) Humans are constantly exposed to a variety of exogenous and endogenous stresses throughout their lives, which cause a variety of responses at the cellular, tissue, organ, and organism levels. Injuries to cellular and tissue functions caused by genetic factors or prolonged stress can accelerate aging.3) Oxidative stress is one of the most important mechanisms of cell aging, causing several age-related diseases. Contributing factors include genomic instability, decreased proteome homeostasis, telomere shortening, changes in stem cell function, cell-to-cell communication defects, mitochondrial dysfunction, and metabolic imbalance.4,5) In recent years, society is rapidly aging its population, and heat stress caused by global warming is seriously affecting the health of the elderly, so it is necessary to prepare for this.6) Aging is an inevitable and multifaceted biological process. The most important factors are reactive oxygen species (ROS) and cell aging. Oxidative stress is defined as damage caused by ROS, by-products of mitochondrial electron transport chains and other major molecular pathways, which can cause cell damage and activate cell aging pathways.7,8) Antioxidants can have a protective effect against oxidative stress and can reduce toxicity. Plant-derived antioxidants have potential antioxidant effects. These antioxidants have positive effects on neurodegenerative diseases, lung fibrosis, atherosclerotic diseases, kidney damage, and liver toxicity caused by oxidants.9) Caenorhabditis elegans has advantages in scientific research like short life cycles, small size, easy maintenance, genetic therapeutic potential, and conserved biological processes associated with aging. C. elegans can be used to efficiently and quickly evaluate compounds that have the potential to slow aging.10)
In the process of finding anti-aging compounds using C. elegans, it was confirmed that the methanol extract of Epimedium koreanum Nakai (Berberidaceae) exhibited long-lived activity. Anti-aging of total flavonoid,11,12) antioxidant of icariin,13) and treatment of Parkinson’s Disease of icaritin14) have been reported in plants in the genus Epimedium. Icarisoside A was isolated by activity-oriented chromatography of E. koreanum.15) In this study, we studied the longevity and thermal shock stress and osmotic stress resistance effects of icarisoside A using the C. elegans model system. Furthermore, we investigated whether ikarisoside A increases heat shock stress resistance protein using mutants (HSP-16.2 expression) with CL2070 transgenic strains. We also investigated whether ikarisoside A can influence aging factors such as pharyngeal pumping, reproduction, movement, and body length. Furthermore, antioxidant capacity of ikarisoside A was analyzed by measuring intracellular ROS level and antioxidant enzyme activity of C. elegans. Then, two mutants (HSP-16.2 and SOD-3 expressions using transgenic strains including CL2070 and CF1553) of stress response genes were used to investigate whether ikarisoside A increases stress tolerance proteins. It was also checked up if ikarisoside A could give influences on aging factors such as pharyngeal pumping, reproduction, length, and body movement. Icarisoside A reduced lipofuscin accumulation in C. elegans.
Materials and Methods
General Materials – The absorbance to examine the antioxidant effects of samples was determined using microplatereader (Sunrise, Grödig, Austria). The fluorescence of GFP-expressing populations was examined under a fluorescence microscope (Olympus, Tokyo, Japan). Agar and 4-hydroxybenzoic acid were purchased from Sigma (USA). Yeast extract and peptone were purchased from BD biosciences (USA).
Extraction and Isolation of Plant Materials–Ikarisoside A from Epimedium koreanum was separated according to the method described previously.15)
C. elegans Strains and Their Maintenance–The worms were grown at 20°C on nematode growth medium (NGM) agar plates in the presence of Escherichia coli strain OP50, as described previously.15)
Lifespan Assay – The wild-type N2 strain of C. elegans was used for lifespan analysis, and the resulting data are representative of at least three independent experiments conducted at 20°C. Embryos from N2 nematodes were isolated and then transferred to NGM plates containing samples to be tested to prepare age-synchronized worms grown with various concentrations of ikarisoside A. These worms were transferred to new NGM plates every 2 days. Worms that did not respond to reproduction with the tip of platinum wires were considered dead.16)
Determination of Thermal and Osmotic Stress Resistances–The age-synchronized nematodes were transferred to fresh plates on day 4 of adulthood and then incubated with or without ikarisoside A at 36°C for the heat stress resistance assays. Worm survival rates were monitored for 25 h.17) To assess the resistance of ikarisoside A to osmotic stress, age-synchronized nematodes were transferred on day 5 of adulthood to NGM agar plates with samples to be tested with 500 mM NaCl, and survival against osmotic stress was measured for 12 h at 20°C.18)
Fluorescence Microscopy and Visualization – The life-extending mechanism of ikarisoside A was investigated using age-synchronized transgenic strain CL2070 containing green fluorescent protein (GFP)-based reporter HSP-16.2::GFP. The CL2070 mutant was heat-shocked at 36°C for 2 h and then recovered at 20°C for 4 h before sample processing. On day 3 of adulthood, transgenic worm strains were anesthetized with sodium azide (4%) and mounted on 2% agarose pads. The GFP fluorescence of the mutant was directly observed with a fluorescence microscope (Olympus, Japan), and protein expression could be assessed using Image J software.19)
Lipofuscin Accumulation – C. elegans was cultured in embryos, as was the life span assay. Fluorescence images of anesthetized worms were taken on day 8 of adulthood using a fluorescence microscope to confirm intestinal lipofuscin levels in nematodes. Pixel intensity values of the worms were extracted from fluorescence microscope images using Image J software.20)
Measurement of Aging-related Factors and Locomotion–The age-synchronized N2 worms were cultured on NGM agar plates with or without ikarisoside A from embryo. During reproductive analysis, worms in the L4 stage were transferred individually to new plates each day to distinguish offspring from their parents. The number of offspring was calculated at L2 or L3 stages.20) In addition, on the fourth and eighth days of adulthood, one worm was transferred to a new plate, and the number of pharyngeal contractions and body movements was counted under an inverted microscope for 20 seconds.20) Furthermore, the worms were photographed for growth change analysis, and each worm's body length was measured in Cellens dimensions (Olympus, Japan).21)
Data Analysis – The data from the lifespan and stress resistance analysis were plotted using the Kaplan-Meier analysis, and statistical significance was determined by a log-rank test. The other data are presented as the mean ± standard deviation or standard error of means (SEM) as indicated. Statistical significance for the differences between treated and control groups was analyzed by the one-way (ANOVA).
Results
Effects of Ikarisoside A on the Lifespan of C. elegans–The ikarisoside A was isolated from the ethyl acetate soluble fraction of the E. koreanum methanol extract as previously described.15) The life-extending activity of ikarisoside A was evaluated in the wild-type N2 worms, which exhibited a concentration-dependent longevity effect (Fig. 1A). As shown in Fig. 1B and Table I, the estimated mean lifespan of the ikarisoside A-treated worm increased significantly by 36.9% (p<0.001) and 50.9% (p<0.001) at 25 μM (15.2 ± 0.3 days) and 50 μM (16.8 ± 0.4 days), respectively.
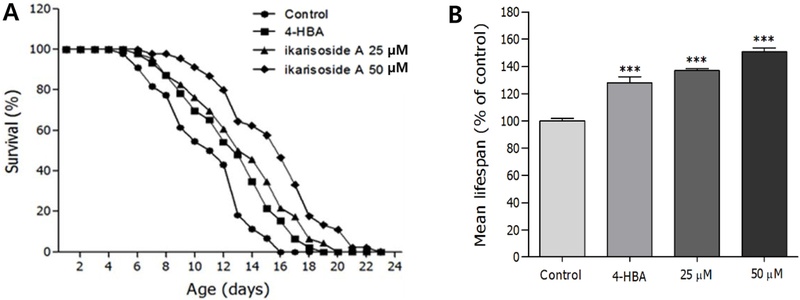
Effects of ikarisoside A from E. koreanum on the lifespan of wild-type N2 nematodes. (A) The mortality of each group was determined by daily counting of surviving and dead worms. (B) The mean lifespan of the N2 worms was calculated from the survival curves. Statistical difference between the curves was analyzed by log-rank test. Error bars represent the standard error of mean (S.E.M.). Differences compared to the control were considered significant at ***p < 0.001 by one-way ANOVA. 4-HBA (4-hydroxybenzoic acid): positive control 25 μM.
Effects of Ikarisoside A on the Stress Tolerance of C. elegans–To investigate the effects of ikarisoside A under stress conditions, osmotic pressure, heat, and oxidative stress were applied to wild-type N2 worms. Resistance to osmotic stress increased by 30% when treated with ikarisoside A at 50 μM compared to untreated controls (Fig. 2A). Furthermore, application of ikarisoside A under thermal stress conditions resulted in 31.1% and 45.5% extended life expectancy of worms at 25 μM (p < 0.001) and 50 μM (p < 0.001), respectively, suggesting that ikarisoside A treatment improved thermal resistance (Fig. 2B, Table II).
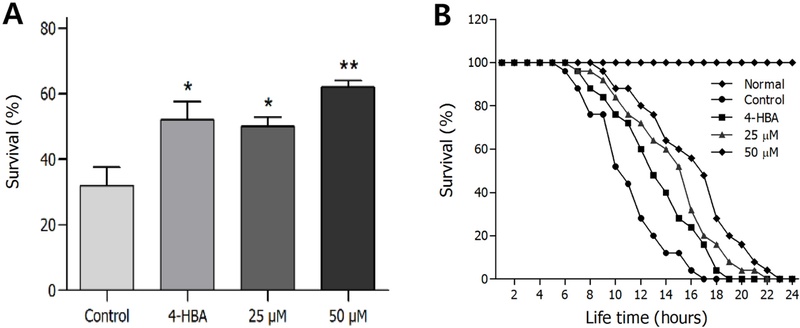
Effects of ikarisoside A from E. koreanum on the stress tolerance of wild-type N2 nematodes. (A) Survival rates under the osmotic stress condition were monitored for 12 h. (B) After incubation of the worms at 36°C, their thermal tolerance was evaluated. Statistical differences between the curves were analyzed by the log-rank test. Error bars represent the standard error of mean (S.E.M.). Differences compared with the control were considered significant at *p < 0.05, and **p < 0.01 by the one-way ANOVA. 4-HBA (4-hydroxybenzoic acid): positive control 25 μM. Normal: untreated worms.
Effects of Ikarisoside A on the HSP-16.2 Expressions in Transgenic Nematodes – To investigate whether the life-extending mechanism of ikarisoside A is associated with increased stress resistance, the transgenic strain CL2070 was used to quantify the expression of protein HSP-16.2. The CL2070 mutant containing the HSP-16.2::GFP reporter gene was subjected to heat shock at 36°C for 2 h, followed by recovery of the worm at 20°C for 4 h, and then the fluorescence intensity was quantified. HSP-16.2::GFP expression levels induced by heat shock were increased by approximately 17.8% by 25 μM of ikarisoside A (p<0.001, Fig. 3A, 3B).
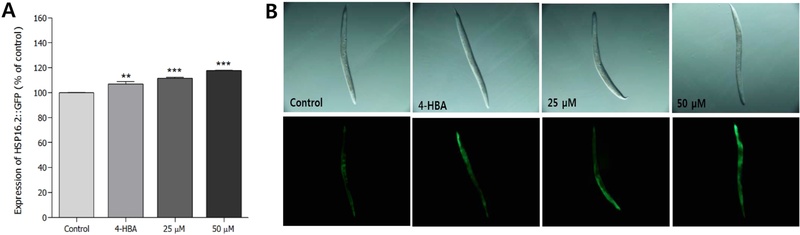
Effects of ikarisoside A from E. koreanum on the expression of HSP-16.2 in C. elegans. (A) The mean GFP intensity of the CL2070 mutant was expressed as the mean ± S.E.M. (B) Images of 16.2::GFP expressions of CL2070 worms were taken from the corresponding mutants grown in the presence or absence of ikarisoside A. Differences compared to the control were considered significant at **p < 0.01 and ***p < 0.01 by one-way ANOVA. 4-HBA (4-hydroxybenzoic acid): positive control 25 μM.
Effects of Ikarisoside A on the Lipofuscin Accumulation – The autofluorescence level of lipofuscin affected by ikarisoside A was measured in C. elegans, showing significant decrease in fluorescence intensity in the ikarisoside A-treated worms by 16.7% at 50 μM, compared to the control (p < 0.001, Fig. 4).
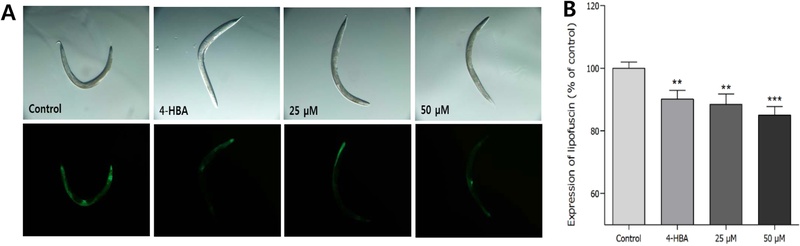
Effects of ikarisoside A from E. koreanum on the lipofuscin accumulation in wild-type N2 nematodes. (A) Image of intestinal autofluorescence from lipofuscin accumulation of worms were taken on the 8th adult days. (B) Fluorescence intensity was measured by determining pixel intensity in worm’s intestines using Image J software. Themean fluorescence intensity of lipofuscin accumulation was expressed as the mean ± S.E.M. Differences compared to the control were considered significant at **p < 0.01 and ***p < 0.001 by one-way ANOVA. 4-HBA (4-hydroxybenzoic acid): positive control.
Effects of Ikarisoside A on the Aging-related Factors of C. elegans and Locomotion – To identify the possible mechanisms by which ikaricoside A affects the lifespan of C. elegans, we calculated the parameter changes of aging-related factors such as progeny, pharyngeal pumping, and body length induced by this ikaricoside A. There were no significant changes in reproduction, food intake, and body length between the ikaricoside A-fed and control worms (Fig. 5A, 5B, and 5C). We also examined the body movements of worms to determine the effect of ikaricoside A on age-related functional changes in C. elegans, but there were no differences between the ikaricoside A-treated worms and control worms (Fig. 5D).
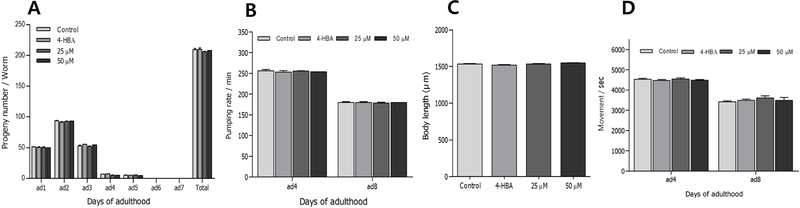
Effects of ikarisoside A from E. koreanum on the various aging-related factors of wild-type N2 nematodes. (A) Daily and total reproductive outputs were counted. The progeny was counted at the L2 or L3 stage. (B) On the 4th and 8th days of adulthood, the pharyngeal pumping rates were measured. (C) For the grown alteration assay, photographs were taken on 4th day of worms and the body length of each animal was analyzed. (D) The body movements were counted on 4th and 8th days of adulthood worms under a dissecting microscope for 20 seconds. Data are expressed as the mean ± S.E.M. of three independent experiments (N=3). 4-HBA (4-hydroxybenzoic acid): positive control 25 μM.
Discussion
The measurement of the life-extending effect of icarisoside A isolated from E. koreanum using C. elegans showed a concentration-dependent longevity effect. It is known that there is a significant correlation between longevity and stress.22) In this study, ikarisoside A was found to increase the survival rate of C. elegans in stressful environments such as heat and osmotic conditions, and resistance to juglone-induced oxidative stress was confirmed in the last report.15) Among them, protein production resistant to heat shock and oxidative stress was confirmed by using the transgenic worms CL2070 and CF1553, respectively.15) The effect of ikarisoside A on the expression levels of SOD-3 and HSP-16.2 genes was evaluated using the transgenic strains CL2070 and CF1553. The worms treated with ikarisoside A had higher GFP intensities than the untreated controls, indicating that ikarisoside A treatment increased the expression levels of SOD-3 and HSP-16.2 genes (Fig. 3A, 3B). And nematodes fed the icarisoside A showed reduced lipofuscin accumulation compared to control worms. In previous studies, we have reported the effects of increasing the activity of antioxidant enzymes SOD and catalase, and inhibiting ROS accumulation in C. elegans of ikarisoside A.15) On the other hand, it did not show any significant changes due to ikarisoside A in aging-related factors such as food intake, exercise, reproduction, and growth. This indicates that ikarisoside A induces longevity activity without affecting these factors. Recently, the effects of ikarisoside on anti-inflammatory, catecholamine secretion, and osteoclast production differentiation inhibitory effects have been reported.23-25) Meanwhile, recent studies related to anti-aging have been reported on icariin, the main component of the same plant.26-28) Icariin and ikarisoside A are flavonoid compounds with similar skeletons, and it is a result of good agreement that ikarisoside A has anti-aging activity.
Conclusions
In summary, taken together with the present study and the preceding study, ikarisoside A extended the life span of, and also increased the expression of osmotic, heat and oxidative stress resistance, and antioxidant enzymes activities. Further efforts to elucidate the mechanism of these effects found that expression of SOD-3 and HSP-16.2 proteins was induced by the ikarisoside A treatment in the mutant of C. elegans. In addition, ikarisoside A reduced intracellular ROS and lipofuscin accumulation. Therefore, ikarisoside A has the potential to be an effective anti-aging compound. To the best of our knowledge, this is the first report on the life-span effect of ikarisoside A. However, the current data are limited data, and further studies are needed to elucidate the clear mechanism of ikarisoside A-mediated life span extending effect.
Acknowledgments
This research was supported by the Regional Innovation System & Education (RISE) program through the Jeonbuk RISE Center, funded by the Ministry of Education (MOE) and the Jeonbuk State, Republic of Korea (2025-RISE-13-WSU).
References
-
Liu, J. K. (2022) Antiaging agents: safe interventions to slow aging and healthy life span extension. Nat. Prod. Bioprospect. 12: 18.
[https://doi.org/10.1007/s13659-022-00339-y]

-
Yang, S. and Lian, G. (2020) ROS and diseases: role in metabolism and energy supply. Mol. Cell Biochem. 467: 1-12.
[https://doi.org/10.1007/s11010-019-03667-9]

-
Raza, U., Tang, X. Liu, Z. and Liu, B. (2024) SIRT7: the seventh key to unlocking the mystery of aging. Physiol. Rev. 104: 253-280.
[https://doi.org/10.1152/physrev.00044.2022]

-
Martemucci, G., Portincasa, P., Di Ciaula, A., Mariano, M., Centonze, V. and D'Alessandro, A. G. (2022) Oxidative stress, aging, antioxidant supplementation and their impact on human health: An overview. Mech. Ageing Dev. 206: 111707.
[https://doi.org/10.1016/j.mad.2022.111707]

-
Galasso, M., Gambino, S., Romanelli, M. G., Donadelli, M. and Scupoli, M. T. (2021) Browsing the oldest antioxidant enzyme: catalase and its multiple regulation in cancer. Free Radic. Biol. Med. 172: 264-272.
[https://doi.org/10.1016/j.freeradbiomed.2021.06.010]

-
Guo, Q., Madaniyazi, L., Nomura, S., Chen, K. and Hashizume, M. (2025) Population aging exacerbates heat stroke-related ambulance transportations in Japan. Environ. Int. 199: 109506.
[https://doi.org/10.1016/j.envint.2025.109506]

-
Almalki, W. H. and Almujri, S. S. (2024) Aging, ROS, and cellular senescence: a trilogy in the progression of liver fibrosis. Biogerontology 26: 10.
[https://doi.org/10.1007/s10522-024-10153-3]

-
Shabalina, I. G., Edgar, D., Gibanova, N., Kalinovich, A. V., Petrovic, N., Vyssokikh, M. Y., Cannon, B., Nedergaard, J. (2024) Enhanced ROS production in mitochondria from prematurely aging mtDNA mutator mice. Biochemistry (Mosc) 89: 279-298.
[https://doi.org/10.1134/S0006297924020081]

-
Omidifar, N., Nili-Ahmadabadi, A., Nakhostin-Ansari, A., Lankarani, K. B., Moghadami, M., Mousavi, S., Hashemi, S. A., Gholami, A., Shokripour, M. and Ebrahimi, Z. (2021) The modulatory potential of herbal antioxidants against oxidative stress and heavy metal pollution: plants against environmental oxidative stress. Environ. Sci. Pollut. Res. Int. 28: 61908-61918.
[https://doi.org/10.1007/s11356-021-16530-6]

-
Chen, X., Bahramimehr, F., Shahhamzehei, N., Fu, H., Lin, S., Wang, H., Li, C., Efferth, T. and Hong, C. (2024) Anti-aging effects of medicinal plants and their rapid screening using the nematode Caenorhabditis elegans. Phytomedicine 129: 155665.
[https://doi.org/10.1016/j.phymed.2024.155665]

-
Wu, B., Xiao, X., Li, S. and Zuo, G. (2019) Transcriptomics and metabonomics of the anti-aging properties of total flavones of Epimedium in relation to lipid metabolism. J. Ethnopharmacol. 229: 73-80.
[https://doi.org/10.1016/j.jep.2018.09.039]

-
Yan, S., Wu, B., Lin, Z., Jin, H., Huang, J., Yang, Y., Zhang, X., Shen, Z. and Zhang, W. (2009) Metabonomic characterization of aging and investigation on the anti-aging effects of total flavones of Epimedium. Mol. Biosyst. 5: 1204-1213.
[https://doi.org/10.1039/b816407j]

-
Yoon, J. W., Lee, S. E., Park, Y. G., Kim, W. J., Park, H. J., Park, C. O., Kim, S. H., Oh, S. H., Lee, D. G., Pyeon, D. B., Kim, E. Y. and Park, S. P. (2021) The antioxidant icariin protects porcine oocytes from age-related damage in vitro. Anim Biosci. 34: 546-557.
[https://doi.org/10.5713/ajas.20.0046]

-
Zheng, C., Wu, L., Luo, L., Cai, J., Huang, Z. and Tian, K. (2025) Therapeutic effects and mechanisms of icaritin in Parkinson's Disease. Curr. Pharm. Des. 31: 1983-1989.
[https://doi.org/10.2174/0113816128344629250115074105]

-
Kim, D. K. and Kim, J. H. (2024) Antioxidant activity of ikarisoside A from Epimedium koranum Nakai in Caenorhabditis elegans. Kor. J. Pharmacogn. 55: 15-21.
[https://doi.org/10.22889/KJP.2024.55.1.15]

-
Bonuccelli, G., Brooks, D. R., Shepherd, S., Sotgia, F. and Lisanti, M. P. (2023) Antibiotics that target mitochondria extend lifespan in C. elegans. Aging (Albany NY) 15: 11764-11781.
[https://doi.org/10.18632/aging.205229]

-
Xu, F., L,i R., von Gromoff, E. D., Drepper, F., Knapp, B., Warscheid, B., Baumeister, R. and Qi, W. (2023) Reprogramming of the transcriptome after heat stress mediates heat hormesis in Caenorhabditis elegans. Nat. Commun. 14: 4176.
[https://doi.org/10.1038/s41467-023-39882-8]

-
Yue, Y., Shen, P., Xu, Y. and Park, Y. (2019) p-Coumaric acid improves oxidative and osmosis stress responses in Caenorhabditis elegans. J. Sci. Food Agric. 99: 1190-1197.
[https://doi.org/10.1002/jsfa.9288]

-
Li, D. Q., Guo, Y. J., Zhang, C. P., Du, H. Z., Hong, Y., Huang, B. S. and Cao, Y. (2021) n-Butanol extract of Hedyotis diffusa protects transgenic Caenorhabditis elegans from Aβ-induced toxicity. Phytother. Res. 35: 1048-1061.
[https://doi.org/10.1002/ptr.6871]

-
Hu, Q., Liu, Z., Guo, Y., Lu, S., Du, H. and Cao, Y. (2021) Antioxidant capacity of flavonoids from Folium Artemisiae Argyi and the molecular mechanism in Caenorhabditis elegans. J. Ethnopharmacol. 279: 114398.
[https://doi.org/10.1016/j.jep.2021.114398]

-
Ji, Y. J., Nam, S., Jin, Y. H., Cha, E. J., Lee, K. S., Choi, K. Y., Song, H. O., Lee, J., Bae, S. C. and Ahnn, J. (2004) RNT-1, the C. elegans homologue of mammalian RUNX transcription factors, regulates body size and male tail development. Dev. Biol. 274: 402-412.
[https://doi.org/10.1016/j.ydbio.2004.07.029]

-
Deepashree, S., Niveditha, S., Shivanandappa, T. and Ramesh, S. R. (2019) Oxidative stress resistance as a factor in aging: evidence from an extended longevity phenotype of Drosophila melanogaster. Biogerontology 20: 497-513.
[https://doi.org/10.1007/s10522-019-09812-7]

-
Choi, H. J., Eun, J. S., Park, Y. R., Kim, D. K., Li, R., Moon, W. S., Park, J. M., Kim, H. S., Cho, N. P., Cho, S. D. and Soh, Y. (2008) Ikarisoside A inhibits inducible nitric oxide synthase in lipopolysaccharide-stimulated RAW 264.7 cells via p38 kinase and nuclear factor-kappaB signaling pathways. Eur. J. Pharmacol. 601: 171-178.
[https://doi.org/10.1016/j.ejphar.2008.09.032]

-
Choi, H. J., Park, Y. R., Nepal, M., Choi, B. Y., Cho, N. P., Choi, S. H., Heo, S. R., Kim, H. S., Yang, M. S. and Soh, Y. (2010) Inhibition of osteoclastogenic differentiation by ikarisoside A in RAW 264.7 cells via JNK and NF-kappaB signaling pathways. Eur. J. Pharmacol. 636: 28-35.
[https://doi.org/10.1016/j.ejphar.2010.03.023]

-
Li, X., Toyohira, Y., Horisita, T., Satoh, N., Takahashi, K., Zhang, H., Iinuma, M., Yoshinaga, Y., Uneo, S, Tsutsui, M., Sata, T. and Yanagihara, N. (2015) Ikarisoside A inhibits acetylcholine-induced catecholamine secretion and synthesis by suppressing nicotinic acetylcholine receptor-ion channels in cultured bovine adrenal medullary cells. Naunyn Schmiedebergs Arch. Pharmacol. 388: 1259-1269.
[https://doi.org/10.1007/s00210-015-1161-y]

-
Li, Y., Wei, Z. F. and Su, L. (2024) Anti-aging effects of icariin and the underlying mechanisms: A mini-review. Aging Med. (Milton) 7: 90-95.
[https://doi.org/10.1002/agm2.12284]

-
Zheng, J., Hu, S., Wang, J., Zhang, X., Yuan, D., Zhang, C., Liu, C., Wang, T. and Zhou, Z. (2021) Icariin improves brain function decline in aging rats by enhancing neuronal autophagy through the AMPK/mTOR/ULK1 pathway. Pharm. Biol. 59: 183-191.
[https://doi.org/10.1080/13880209.2021.1878238]

-
Sun, J., Lv, C., Lin, Z., Luo, S., Liao, S., Lian, X., Liu, Y., Luo, L., Zheng, M., Zhang, D., Wang, X. and Wang, S. (2025) Integrating network pharmacology and experimental validation to decipher the protective effect of icariin against testicular aging. Ecotoxicol. Environ. Saf. 303: 119063.
[https://doi.org/10.1016/j.ecoenv.2025.119063]

